Blog
Why multi-drug panels matter: compliance and safety

TL;DR:
- Basic drug tests miss 69% of polysubstance positives, risking undetected employee use.
- Multi-drug panels detect a broader range of substances, improving accuracy and compliance.
- Upgrading to comprehensive panels reduces false positives and better addresses modern substance use trends.
Most workplace drug testing programs were built around a simple idea: run a basic panel, get a result, move on. But that logic has a serious flaw. Immunoassay misses 69% of polysubstance positives compared to definitive testing methods, meaning a significant share of at-risk employees can walk right through your screening process undetected. Today’s substance landscape includes fentanyl analogs, synthetic cannabinoids, and designer opioids that simply do not show up on a standard 5-panel test. This guide walks you through why multi-drug panels exist, what the data actually says, and how to build a testing approach that holds up to modern compliance demands.
Key Takeaways
| Point | Details |
|---|---|
| Comprehensive detection | Multi-drug panels reveal a much broader range of substance use, slashing undetected cases. |
| Lower compliance risk | Advanced panels help workplaces meet tough regulatory standards and prevent legal gaps. |
| Cost-effective safety | Paying slightly more for bigger panels can avert expensive incidents and protect your team. |
| Data-driven decisions | Use real outcomes and workplace data to continuously inform your testing approach. |
What are multi-drug panels and why do they matter?
A multi-drug panel is a single test that screens for multiple substances at once. Instead of checking for five common drugs, advanced panels can screen for 12, 15, 18, or even 22 or more substances in one urine sample. That shift from basic to broad coverage is not just a product upgrade. It reflects a fundamental change in how substances are used and abused in real workplaces.
Basic 4 and 5-panel tests were designed for a different era. They typically detect THC, cocaine, opiates, amphetamines, and PCP. That lineup made sense decades ago. Today, it leaves enormous blind spots. Synthetic opioids, benzodiazepines, barbiturates, methamphetamine variants, and newer designer drugs are not captured by those legacy panels.
Multi-panels are proven superior for harm reduction in the current polysubstance era, where individuals often use more than one substance simultaneously. Missing one substance in a combination can completely distort your risk picture.
Here is what a more advanced panel adds over a basic 5-panel test:
- Fentanyl and fentanyl analogs (increasingly present in workplace incidents)
- Synthetic cannabinoids (K2, Spice) that do not trigger standard THC cutoffs
- Benzodiazepines (Xanax, Valium) widely misused but absent from basic panels
- Buprenorphine and tramadol (prescription opioids often overlooked)
- Ethyl glucuronide (EtG) for alcohol metabolite detection
- Kratom (KRA) and other emerging botanical substances
For a deeper look at how these panels are structured, what is multi-drug panel testing provides a useful breakdown of detection categories and cutoff thresholds.
Pro Tip: When reviewing your panel options, pay specific attention to whether synthetic cannabinoids and fentanyl are included. These are the two categories most likely to be missed by basic panels and most likely to be present in high-risk environments.
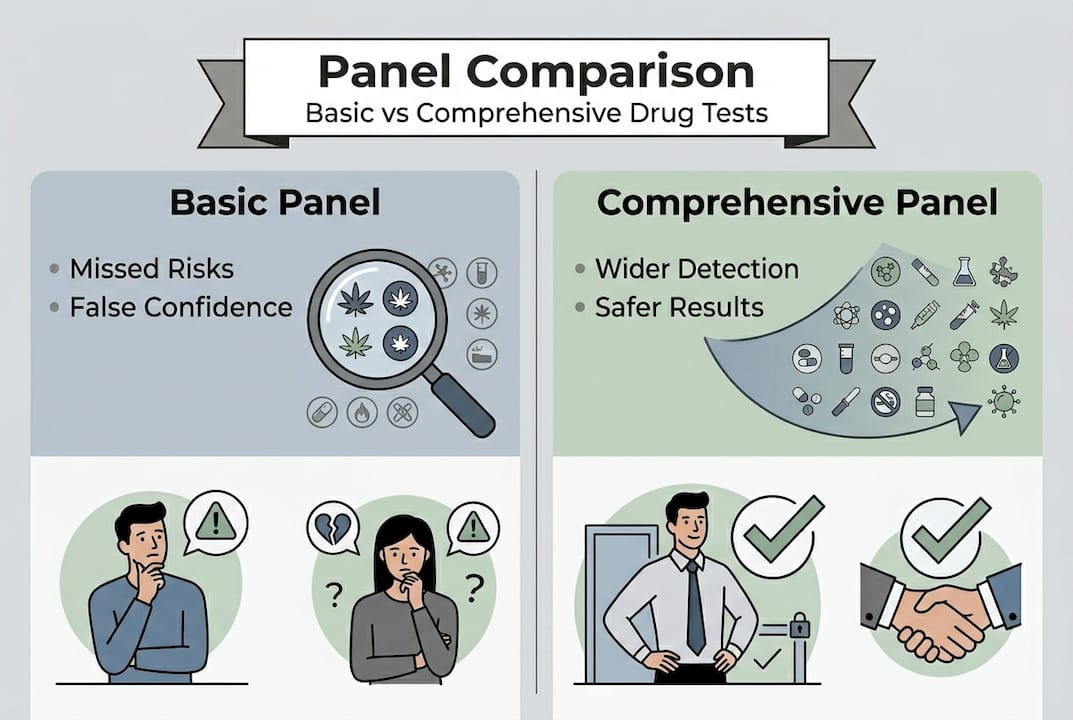
Common limitations of basic panels: False reassurance and missed risk
The danger with basic panels is not just what they miss. It is the false confidence they create. A negative result on a 5-panel test feels like a clean bill of health. But if the employee is using fentanyl, a synthetic opioid, or a benzodiazepine, that result tells you nothing useful.
Basic 4 and 5-panel tests are cheaper but miss emerging drugs and create a false sense of reassurance for organizations that believe their testing program is thorough. The cost savings are real, but so is the exposure.
Consider what falls outside a standard DOT 5-panel screen:
- Fentanyl (responsible for the majority of overdose deaths in the U.S.)
- Synthetic cannabinoids (unpredictable potency, no THC cross-reactivity)
- Benzodiazepines beyond a narrow opiate window
- Gabapentin and pregabalin (increasingly misused, especially alongside opioids)
- Designer stimulants (bath salts, cathinones)
For organizations operating in transportation, construction, healthcare, or manufacturing, any one of these substances in an employee’s system represents a serious liability. A single workplace incident tied to a substance your panel does not detect can result in regulatory action, litigation, and reputational damage that far exceeds years of savings from cheaper tests.
“The DOT 5-panel standard was built for a different drug landscape. For high-risk industries dealing with today’s opioid and synthetic drug crisis, 14 to 28-panel testing is no longer optional. It is the baseline for responsible compliance.”
The workplace harm reduction framework reinforces this point: detection is only meaningful when it covers the substances your workforce is actually exposed to. Anything less is a gap in your program, not a feature.
The science behind enhanced detection: Data shows why comprehensive panels work
Let’s look at the numbers directly. A peer-reviewed study comparing immunoassay screening to definitive mass spectrometry (MS) testing found that MS identified 69% more positives than standard immunoassay panels. Across 883 patients, MS produced 7,086 additional results that required clinical or compliance intervention. That is not a marginal difference. That is a structural failure in basic screening.
The same data showed that advanced testing also eliminated 14.6% of false positives that immunoassay had flagged incorrectly. So comprehensive panels do not just catch more. They catch more accurately.
| Metric | Immunoassay (basic) | Definitive MS (advanced) |
|---|---|---|
| Additional positives detected | Baseline | +69% |
| False positive rate | Higher | Reduced by 14.6% |
| Polysubstance detection | Limited | Significantly improved |
| Substances covered | 4 to 5 | 12 to 28+ |
This matters for compliance officers because false positives carry their own risks. An employee wrongly flagged faces unnecessary consequences, and your organization faces potential legal exposure. Comprehensive panels reduce both ends of the error spectrum.
For organizations building or updating their testing programs, the multi-panel compliance guide outlines how to align panel selection with both detection accuracy and regulatory requirements.
Key takeaway: The cost of a missed positive is not just a safety incident. It is a compliance failure, a potential legal event, and a signal that your program is not actually working.
Choosing the right panel for your workplace: Practical considerations
Not every organization needs the same panel. A corporate office environment carries different risk profiles than a construction site or a long-haul trucking operation. The right panel is the one that matches your industry regulations, your workforce’s substance exposure, and your compliance obligations.
Higher panels (12 or more) offer better risk mitigation despite a slight cost increase per test. For most safety-sensitive environments, that trade-off is straightforward.
| Panel size | Substances detected | Best use case |
|---|---|---|
| 5-panel | THC, cocaine, opiates, amphetamines, PCP | Low-risk office settings, basic compliance |
| 12-panel | Adds benzos, methadone, oxycodone, barbiturates | General workplace, moderate risk |
| 15-panel | Adds buprenorphine, tramadol, fentanyl | Healthcare, transportation, manufacturing |
| 18-panel | Adds K2, KRA, EtG, additional synthetics | High-risk industries, harm reduction programs |
| 22 or more panels | Full synthetic and designer drug coverage | Forensic, government, maximum compliance |
Here is a practical process for selecting your panel:
- Audit your regulatory requirements. DOT, SAMHSA, state laws, and industry-specific rules may define minimum panel requirements.
- Assess your workforce risk profile. Consider the substances most prevalent in your region and industry.
- Review incident history. Past workplace accidents or near-misses involving substance use can reveal detection gaps.
- Match panel to risk level. Use the table above as a starting framework, then adjust based on your audit.
- Consult your legal and HR teams. Ensure panel selection aligns with your written drug-free workplace policy.
For a detailed breakdown of available configurations, the panel options guide covers every panel size from 5 to 28 with substance-by-substance comparisons.
Pro Tip: Set a calendar reminder to review your panel selection every 12 months. New synthetic drugs emerge regularly, and what was cutting-edge coverage in 2024 may already have gaps in 2026.
Implementing and optimizing multi-drug testing in your organization
Selecting the right panel is step one. Getting it embedded into your organization’s daily operations is where programs succeed or fail. A strong multi-drug testing rollout requires policy, training, communication, and a feedback loop that improves over time.
Multi-drug panels are essential tools for compliance and harm reduction strategies, but only when they are backed by a policy that specifies how results are used, who reviews them, and what actions follow.
Follow this rollout sequence:
- Update your written policy. Specify which panel is used, under what circumstances testing occurs, and how results are handled. Be explicit about which substances are covered.
- Train your supervisors and HR team. They need to understand what the panel detects, what a positive result means, and what the escalation process looks like.
- Communicate clearly with employees. Transparency about what is being tested reduces legal challenges and builds trust in the program.
- Establish a review cycle. Quarterly data reviews help you spot trends, such as a spike in a particular substance, before they become incidents.
- Document everything. Chain of custody, result handling, and follow-up actions must be recorded consistently.
Common pitfalls include using panels that do not match your policy language, failing to train supervisors on result interpretation, and skipping annual reviews as the substance landscape shifts.
The compliance best practices resource covers current industry standards and documentation requirements that support defensible, audit-ready programs.
Pro Tip: Use your testing data as a management tool, not just a compliance checkbox. Trends in positive results across departments or job sites can point to systemic issues that need targeted intervention.
Why basic panels leave you exposed: An expert perspective
There is a persistent belief in workplace safety circles that the cheapest test that meets minimum requirements is the smart financial choice. We disagree, and the data backs us up.
The real cost of a basic panel is not the price per test. It is the cost of the incident that happens because your test did not catch what it needed to catch. One workplace injury tied to an undetected fentanyl or synthetic cannabinoid use can generate workers’ compensation claims, OSHA investigations, litigation, and reputational damage that dwarfs years of savings from budget testing.
Immunoassay limitations underscore why high-stakes environments need definitive testing. The gap between what basic panels detect and what employees are actually using has never been wider than it is right now.
We have seen organizations upgrade from 5-panel to 12 or 18-panel testing and immediately identify positive cases they had been missing for years. That is not a testing problem. That is a risk management failure that was hiding in plain sight.
For a broader view of what effective harm reduction looks like in practice, harm reduction insights offers real-world context for building programs that actually work.
The question is not whether you can afford comprehensive panels. It is whether you can afford not to use them.
Get started with reliable multi-drug panel solutions
If your current testing program relies on a basic 5-panel cup, it is time to close the gap. Buy Test Cup offers a full range of advanced multi-drug panel solutions designed for compliance-focused workplaces. The 12 panel test cup is a strong starting point for organizations moving beyond legacy panels, while the 18 panel test cup covers fentanyl, K2, kratom, EtG, and adulterant detection in a single CLIA-waived cup. Both are available for bulk ordering with same-day shipping options. Browse all drug test cup options to find the panel configuration that fits your risk profile and compliance requirements.
Frequently asked questions
What is the main advantage of multi-drug panel testing over single-panel or basic panels?
Multi-drug panels detect a wider range of substances in one test, dramatically reducing missed positives and giving compliance officers a far more accurate picture of workplace substance use. Multi-panels are proven superior for polysubstance harm reduction compared to basic immunoassay screens.
How do multi-drug panels help with legal and regulatory compliance?
Broader detection helps organizations satisfy DOT, state, and federal requirements by capturing synthetic opioids, designer drugs, and emerging substances that single panels routinely miss. Higher panels (12 or more) offer better risk mitigation for organizations with safety-sensitive roles.
Are advanced multi-drug panels more expensive?
Advanced panels cost slightly more per test, but higher panels offer better risk mitigation and the prevention of a single incident or regulatory action easily offsets the price difference over time.
How often should organizations update their testing panels?
Yearly reviews are the standard recommendation, since new threats emerge regularly and panels that were adequate last year may already have blind spots given the pace of synthetic drug development.