Blog
Guide to Multi-Panel Drug Testing for Compliance Success

Finding the right multi-panel drug testing strategy gets complicated when balancing workplace safety and strict regulatory demands. Compliance officers at United States government agencies know that regulatory compliance is not just a legal requirement—it is a safeguard for employee well-being and public trust. This guide walks through the major steps, from assessing organizational testing needs to securing accurate results, highlighting best practices that directly address the unique compliance landscape facing American institutions.
Step 1: Assess testing requirements for your organization
Designing a comprehensive multi-panel drug testing strategy requires careful evaluation of your organization’s unique compliance landscape. Workplace safety demands a systematic approach to understanding your specific testing needs.
To effectively assess your testing requirements, start by examining several critical factors:
- Industry regulations: Different sectors have distinct drug testing mandates
- Federal and state compliance standards for your specific workplace
- Potential safety risks inherent in your work environment
- Job roles and associated safety-sensitive positions
- Frequency of testing needed for various employee categories
The Federal Motor Carrier Safety Administration (FMCSA) provides clear guidance for organizations with commercial driver license (CDL) holders. Regulatory compliance often starts with understanding which substances you need to test for and how frequently.
A well-designed drug testing program protects both employee safety and organizational integrity.
Specific considerations include determining the appropriate multi-panel drug test configuration that matches your organizational risks. Some companies might require 5-panel tests covering core substances, while others need more extensive 10-panel screenings that detect a broader range of controlled substances.
The British Safety Council recommends tailoring your drug testing policy to your specific workplace risks, ensuring that your approach supports both safety objectives and legal requirements.
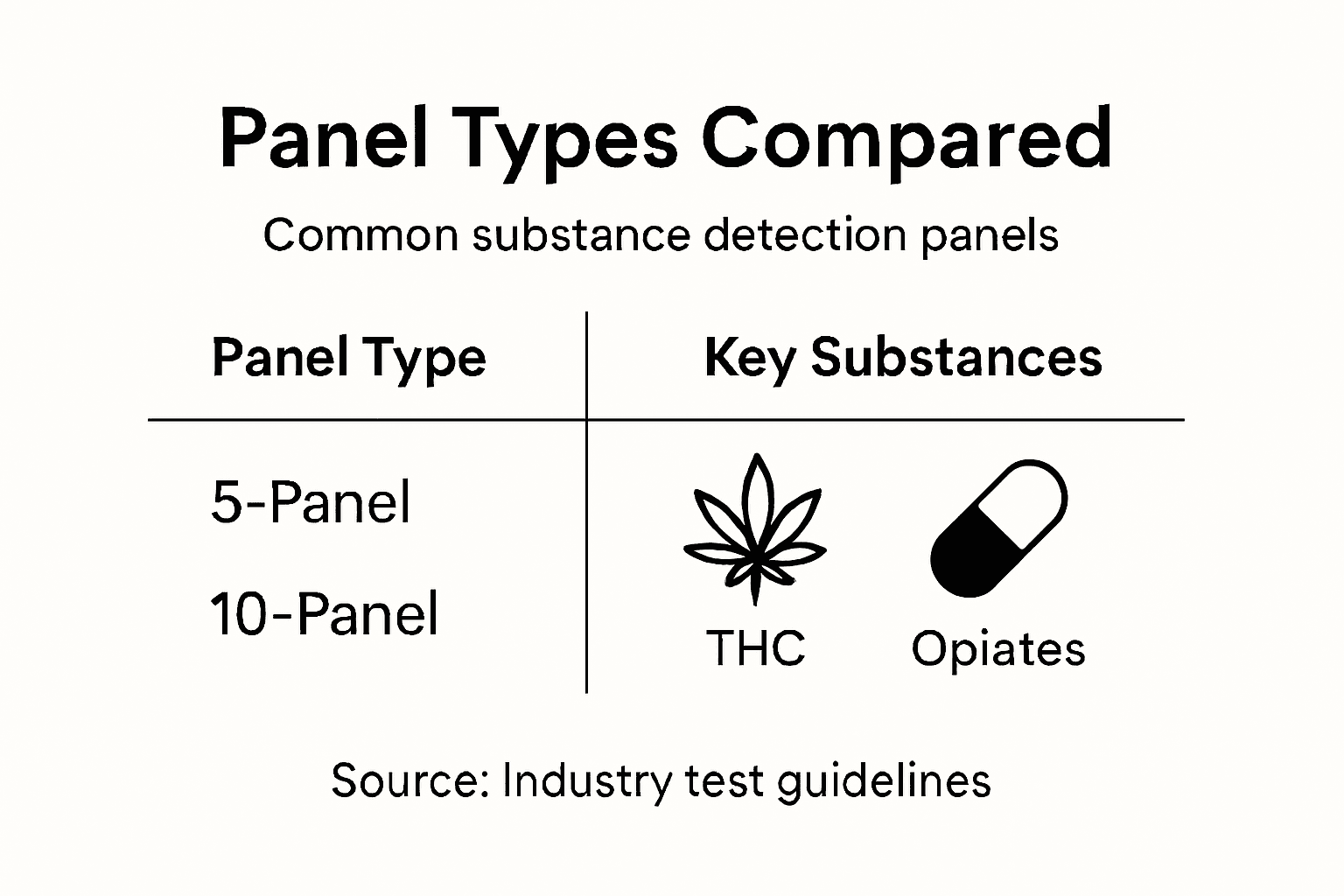
Here’s a comparison of common multi-panel drug test configurations and their typical applications:
| Panel Type | Substances Detected | Typical Use Case |
|---|---|---|
| 5-Panel | THC, cocaine, opiates, amphetamines, PCP | DOT compliance, standard workplace |
| 7-Panel | Adds barbiturates, benzodiazepines | Transportation, safety-sensitive sectors |
| 10-Panel | Includes methadone, propoxyphene, Quaaludes | Healthcare, law enforcement |
| 12-Panel | Broader, adds synthetic drugs | High-risk industries, specialized roles |
Pro tip: Consult with a legal expert specializing in workplace drug testing to ensure your screening program meets all current regulatory standards and protects your organization from potential compliance vulnerabilities.
Step 2: Select appropriate multi-panel drug testing kits
Choosing the right multi-panel drug testing kit is crucial for maintaining workplace safety and regulatory compliance. Your selection will directly impact the effectiveness of your screening program and the protection of your organization.
When selecting a multi-panel drug testing kit, consider these key factors:
- Detection panel range: From 5 to 13 panel configurations
- Specific substances relevant to your industry
- Accuracy and reliability of the testing method
- Compliance with federal and state regulations
- Cost-effectiveness of the screening solution
The American Society of Addiction Medicine (ASAM) recommends carefully evaluating the biological samples and testing methodologies that best suit your organizational needs. Different work environments require different levels of screening complexity.
Selecting the right drug testing kit is more than a compliance checkbox – it’s a critical safety investment.
Specific considerations include understanding the sensitivity and specificity of various testing methods. Some organizations might require drug test cup types that provide comprehensive screening, while others need more targeted approaches.
Advanced multi-panel kits can detect a wide range of substances, from traditional illicit drugs to newer synthetic compounds. Consider the specific risks in your workplace when determining the appropriate panel configuration.
Pro tip: Consult with a drug testing compliance expert to ensure your selected kit meets all current regulatory standards and provides the most accurate screening possible for your specific industry requirements.
Step 3: Set up secure specimen collection procedures
Establishing robust specimen collection procedures is critical for maintaining the integrity and reliability of your drug testing program. Your approach will directly impact the legal defensibility and accuracy of test results.
Key components of a secure specimen collection process include:
- Strict chain of custody documentation
- Designated secure collection areas
- Trained collection personnel
- Tamper-evident collection containers
- Comprehensive documentation protocols
The University of South Florida’s specimen management guidelines emphasize the importance of comprehensive training for all personnel involved in the collection process. Every individual must understand biosafety protocols and chain-of-custody requirements.
Specimen collection is not just a procedure – it’s a critical link in maintaining workplace safety and legal compliance.
Important steps for implementing secure collection include:
- Designate a controlled collection area with limited access
- Require photo identification for all tested individuals
- Implement strict privacy and dignity protocols
- Use tamper-evident collection kits
- Maintain detailed documentation at every stage
The World Anti-Doping Agency recommends developing comprehensive standard operating procedures that minimize the potential for sample contamination or tampering. This involves careful selection of collection equipment and establishing clear guidelines for handling and transport.
Pro tip: Conduct periodic training and simulation exercises to ensure your collection team remains sharp and consistent in following secure specimen handling protocols.
Step 4: Administer and interpret drug test panels
Effective drug test administration and interpretation require precision, clinical knowledge, and a systematic approach to ensure accurate and legally defensible results. Your ability to correctly manage this process will directly impact workplace safety and regulatory compliance.
When administering drug test panels, focus on these critical elements:
- Proper specimen collection techniques
- Consistent testing methodology
- Careful chain of custody documentation
- Understanding potential cross-reactivity
- Accurate result interpretation
The American Society of Addiction Medicine (ASAM) provides comprehensive guidelines for navigating the complexities of drug testing. Pharmacological understanding is crucial for distinguishing between legitimate medical prescriptions and potential substance misuse.
Accurate drug test interpretation is more than reading results – it’s about understanding the nuanced context of each screening.
Key steps for precise test panel administration include:
- Verify donor identification
- Follow strict collection protocol
- Document every stage of testing
- Use calibrated testing equipment
- Analyze results with clinical context
Mayo Clinic research emphasizes the importance of recognizing potential false positives and understanding the intricate biochemical interactions that can influence test outcomes. This requires a sophisticated approach that goes beyond simple positive or negative classifications.
Pro tip: Develop a standardized review process that involves a medical review officer to validate results and provide an additional layer of professional interpretation.
Step 5: Verify test results and ensure regulatory compliance
Ensuring the accuracy and legal defensibility of drug test results requires a meticulous approach to verification and compliance. Your organization’s ability to systematically validate test outcomes will protect both workplace safety and legal standing.
Critical elements of result verification include:
- Comprehensive documentation protocols
- Independent result confirmation
- Strict quality control measures
- Detailed record maintenance
- Regulatory standard alignment
The Food and Drug Administration (FDA) provides comprehensive guidelines for maintaining quality-controlled environments that support accurate testing and result verification. These standards are designed to ensure consistent and reliable screening processes.
Regulatory compliance is not a checkbox – it’s a continuous commitment to precision and integrity.
Key steps for thorough result verification:
- Conduct independent review by qualified personnel
- Cross-reference initial test results
- Validate testing equipment calibration
- Document all verification processes
- Maintain secure and confidential records
The European Union’s Good Manufacturing Practice (GMP) guidelines emphasize the importance of data integrity and personnel competence in maintaining rigorous testing standards. This requires a systematic approach that goes beyond simple result reporting.
The following table summarizes key roles in maintaining effective drug testing program integrity:
| Role | Primary Responsibility | Value to Program |
|---|---|---|
| Collection Personnel | Secure specimen handling | Ensures legal compliance |
| Compliance Officer | Regulatory oversight | Reduces risk of penalties |
| Medical Review Officer | Result validation and review | Adds expert interpretation |
| Quality Control Staff | Equipment and process audits | Maintains data accuracy |
Pro tip: Implement a robust medical review officer process that provides an additional layer of professional scrutiny and interpretation for all drug test results.
Enhance Your Multi-Panel Drug Testing Program with Trusted Supplies
Designing a compliant multi-panel drug testing strategy involves selecting the right tests, ensuring secure specimen collection, and verifying results with precision. If you are seeking reliable products that support accuracy and regulatory compliance, Buy Test Cup offers a comprehensive range of drug testing supplies tailored to your specific needs. From multi-panel drug test cups to tamper-evident specimen collection kits, our products help you maintain workplace safety and meet federal requirements with confidence.
Start building a fully compliant and effective drug screening program today by exploring our extensive catalog designed for government agencies, workplaces, and clinics. Don’t wait to protect your organization and employees—visit Buy Test Cup now and find the testing solutions that fit your regulatory compliance goals.
Frequently Asked Questions
What factors should we consider when assessing our multi-panel drug testing requirements?
Assess your organization’s unique compliance landscape by examining industry regulations, federal and state standards, and potential safety risks in your work environment. Identify job roles that may require testing and determine the frequency needed for various employee categories to tailor your testing strategy effectively.
How do I choose the right multi-panel drug testing kit for our organization?
Select a multi-panel drug testing kit by considering the specific substances relevant to your industry, the detection panel range, and the accuracy of the testing method. Compare different kits for their cost-effectiveness and compliance with applicable regulations to ensure optimal safety and reliability.
What are the best practices for secure specimen collection in drug testing?
Implement a secure specimen collection process by establishing a controlled collection area, using tamper-evident containers, and ensuring thorough training for collection personnel. Maintain strict chain-of-custody documentation to uphold the integrity and reliability of your drug testing program.
How can we effectively administer and interpret drug test panels?
Administer drug test panels with precision by following proper specimen collection techniques and maintaining accurate chain-of-custody documentation. Ensure a qualified medical review officer interprets results for context and understanding, which will enhance the accuracy of your testing outcomes.
What steps should we take to verify drug test results for compliance?
Verify drug test results by conducting an independent review, calibrating testing equipment, and maintaining comprehensive documentation protocols. Implement strict quality control measures to ensure consistent and reliable outcomes, safeguarding your organization against compliance risks.
How can a medical review officer enhance our multi-panel drug testing program?
A medical review officer validates and interprets drug test results, adding a layer of expert oversight that considers legitimate medical prescriptions and potential substance misuse. Establish a standardized review process that incorporates this role to bolster the accuracy and reliability of your drug testing program.