Blog
Drug testing terminology guide for professionals 2026

You might assume drug testing terminology is straightforward, but the reality is far more complex. Misunderstanding terms like cutoff concentration, cross-reactivity, or confirmatory testing can lead to compliance failures, legal exposure, and inaccurate interpretations that affect workplace safety and employee rights. Government agencies, healthcare facilities, and corporate compliance officers navigate a landscape where federal regulations, evolving substance use patterns, and legal obligations intersect with technical laboratory language. This guide clarifies essential drug testing terminology, helping you confidently implement programs that meet regulatory standards while protecting your organization from costly mistakes.
Key takeaways
| Point | Details |
|---|---|
| Testing methods | Immunoassays provide rapid screening while mass spectrometry confirms results with legal certainty |
| Federal regulations | 42 CFR and 49 CFR define drug panels, cutoffs, and fentanyl inclusion requirements for 2026 |
| Legal compliance | ADA mandates reasonable accommodations for disability-related drug testing before and after hiring |
| Interpretation challenges | False positives, detection windows, and clinical relevance complicate result accuracy |
| Evolving terminology | Rising fentanyl and synthetic drug prevalence drives updates to testing vocabulary and protocols |
Understanding core drug testing methods and terminology
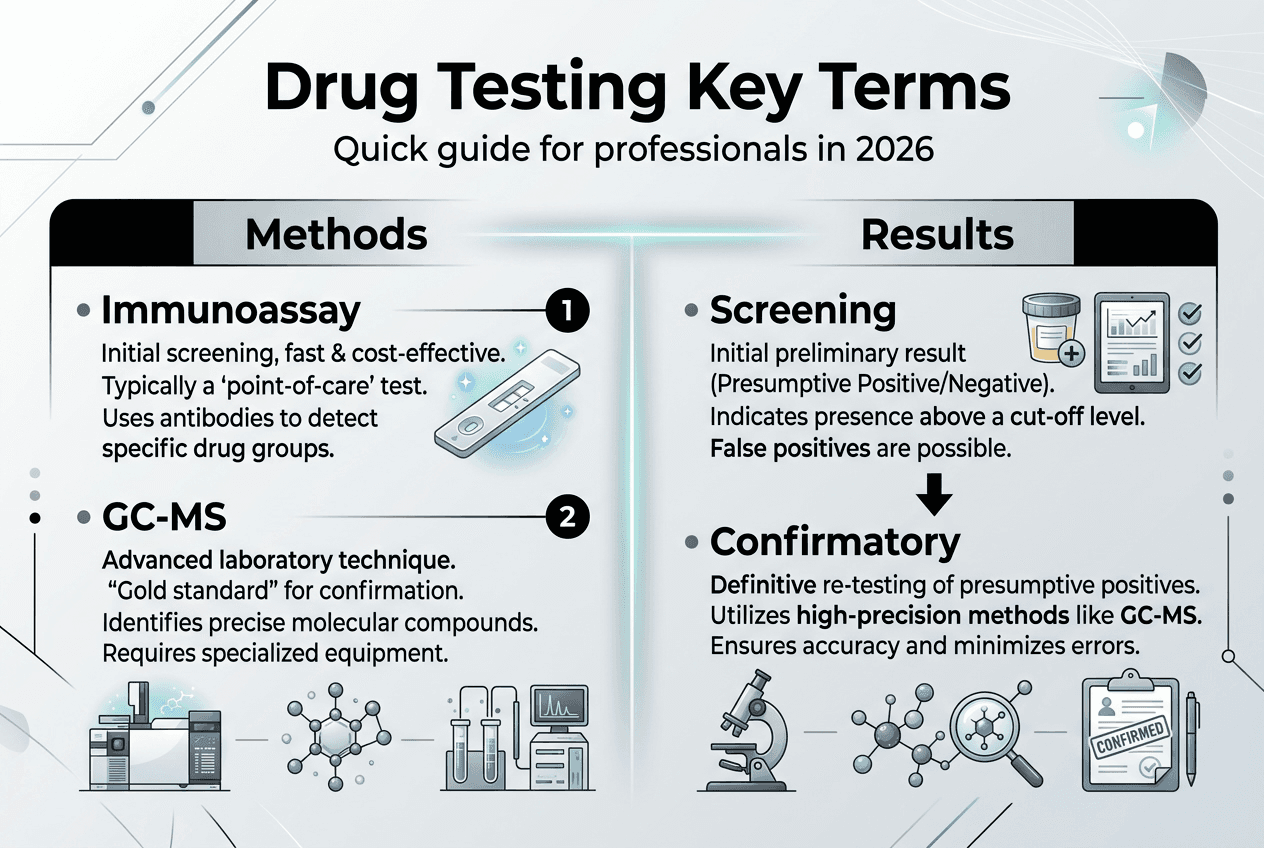
Drug testing relies on distinct methodologies that determine how quickly you receive results and how defensible those results are in legal proceedings. Toxicology screening uses immunoassays for rapid detection and chromatographic or mass spectrometric techniques for confirmatory testing. Understanding these methods helps you interpret terminology correctly and select appropriate tests for your compliance needs.
Immunoassays serve as the frontline screening tool in most workplace and clinical settings. These tests use antibodies that bind to drug molecules, producing a color change or other signal when substances exceed predetermined cutoff concentrations. You get results in minutes, making immunoassays ideal for point-of-care testing and high-volume screening. However, immunoassays can produce false positives due to cross-reactivity, where structurally similar compounds trigger positive results even when the target drug is absent.
Confirmatory testing employs gas chromatography-mass spectrometry (GC-MS) or liquid chromatography-tandem mass spectrometry (LC-MS/MS) to verify presumptive positive immunoassay results. These techniques separate drug molecules and measure their mass-to-charge ratios with extreme precision, eliminating cross-reactivity concerns. Confirmatory tests provide the legal certainty required for employment termination, criminal prosecution, or medical treatment decisions. The terminology distinction between screening and confirmatory testing matters because only confirmed results should trigger adverse actions.
Detection windows describe how long after use a drug remains detectable in biological specimens. Cannabis may appear in urine for weeks in chronic users, while cocaine typically clears within days. Sensitivity refers to a test’s ability to correctly identify true positives, while specificity measures its ability to correctly identify true negatives. High sensitivity reduces false negatives but may increase false positives, requiring you to balance these characteristics based on your program’s goals.
Pro Tip: Always specify whether you’re discussing screening or confirmatory results when communicating with stakeholders. This simple terminology distinction prevents confusion about result reliability and legal defensibility.
Understanding drug testing methods comparison helps you select appropriate technologies for different testing scenarios. Oral fluid testing offers shorter detection windows but easier collection, while hair testing provides months of detection history but higher costs. Each method carries unique terminology related to collection procedures, cutoff levels, and interpretation standards that you must master for effective program management.
Key regulatory frameworks and evolving drug panels for compliant testing
Federal regulations establish the foundation for drug testing terminology by defining which substances to test, at what concentrations, and using which methodologies. Federal regulations at 42 CFR Chapter 1 and 49 CFR part 40 provide frameworks that govern drug testing panels and cutoff concentrations. These regulations directly influence the terminology you encounter in laboratory reports, test kit specifications, and compliance documentation.
The Department of Health and Human Services (HHS) guidelines at 42 CFR establish the five-panel drug test standard for federal workplace testing, covering marijuana, cocaine, opiates, amphetamines, and phencyclidine. The Department of Transportation (DOT) regulations at 49 CFR part 40 apply similar standards to safety-sensitive transportation positions. Both frameworks specify initial screening cutoffs and confirmatory testing cutoffs, creating two-tiered terminology that distinguishes presumptive from definitive results.
Fentanyl and norfentanyl joined regulated panels in 2026 due to overdose prevalence, fundamentally changing drug testing terminology and panel configurations. This addition reflects the evolving threat landscape and requires you to update procurement specifications, laboratory contracts, and result interpretation protocols. Fentanyl’s potency means effective detection requires lower cutoff concentrations than traditional opiates, introducing new terminology around nanogram-per-milliliter thresholds.
| Drug Class | Screening Cutoff (ng/mL) | Confirmatory Cutoff (ng/mL) | Regulatory Authority |
|---|---|---|---|
| Marijuana (THC) | 50 | 15 | 42 CFR, 49 CFR |
| Cocaine | 150 | 100 | 42 CFR, 49 CFR |
| Opiates | 2000 | 2000 | 42 CFR, 49 CFR |
| Amphetamines | 500 | 250 | 42 CFR, 49 CFR |
| Fentanyl | 1 | 1 | 42 CFR (2026) |
| Norfentanyl | 1 | 1 | 42 CFR (2026) |
Cross-reactivity requirements for fentanyl immunoassays present unique challenges because structurally similar synthetic opioids may trigger positive results. You must understand terminology distinguishing between the parent compound (fentanyl) and its metabolite (norfentanyl), as well as analogs like carfentanil or acetylfentanyl. Regulatory definitions affect how laboratories report results and how you document findings in personnel files or medical records.
Understanding these frameworks helps you navigate drug test cups product specifications and ensure your procurement aligns with applicable regulations. Test cups labeled as DOT-compliant or HHS-certified use specific terminology indicating they meet federal cutoff requirements and quality standards. Mismatching test specifications to regulatory requirements creates compliance gaps that audits or legal challenges will expose.
Clinical interpretation challenges and medicolegal considerations in drug testing
Interpreting drug test results requires navigating technical limitations, biological variability, and legal obligations that extend beyond simple positive or negative determinations. Test interpretation faces challenges from variability in detection thresholds, accuracy, and clinical relevance, with false positives, false negatives, and narrow detection windows complicating decision-making. You must understand these limitations to avoid overconfidence in test results and ensure fair treatment of tested individuals.
Common interpretation challenges include:
- False positives from cross-reactive substances like certain medications, supplements, or foods that trigger immunoassay responses
- False negatives when drug concentrations fall below cutoff thresholds despite recent use, particularly with diluted specimens
- Detection window variability based on metabolism, hydration, frequency of use, and individual physiology
- Passive exposure scenarios where environmental contact produces low-level positive results without intentional use
- Adulterant interference from substances added to specimens to mask drug presence or invalidate testing
Medicolegal terminology becomes critical when test results inform employment decisions, custody determinations, or criminal proceedings. Chain of custody documentation uses specific language describing specimen collection, handling, storage, and analysis to ensure result integrity. Medical review officer (MRO) terminology describes the physician role in reviewing positive results, interviewing donors, and determining whether legitimate medical explanations exist for positive findings.
Employers must provide reasonable accommodations under the ADA for applicants with disabilities, including those related to drug testing. This obligation introduces terminology around the interactive process, where you engage with applicants to identify and implement accommodations that enable testing without violating disability rights. Accommodations might include modified collection procedures for individuals with physical limitations, extended timeframes for individuals with anxiety disorders, or alternative specimen types for those unable to provide urine samples.
Pro Tip: Document every step of the accommodation process using precise terminology that demonstrates good faith engagement. Vague documentation like “discussed options” provides no legal protection compared to detailed notes specifying requested accommodations, feasibility assessments, and implemented solutions.
Ignoring disability accommodation requirements in drug testing policies exposes organizations to EEOC complaints, litigation, and reputational damage that far exceeds the administrative burden of implementing reasonable modifications to standard procedures.
Understanding these interpretation challenges helps you implement drug tests types for compliance that balance detection capabilities with fairness and legal defensibility. When fentanyl testing became mandatory, many organizations adopted fentanyl test strip details with 1 ng/mL sensitivity to ensure detection of this potent opioid. The terminology around nanogram sensitivity matters because it communicates the test’s ability to detect extremely low concentrations that still indicate use.
Trends in substance use and implications for terminology in workplace drug testing
Substance use patterns evolve constantly, forcing drug testing terminology and protocols to adapt. Encounters related to opioids, cocaine, marijuana, and synthetic drugs increased significantly over three decades, reshaping testing priorities and introducing new terminology around emerging threats. Understanding these trends helps you anticipate terminology changes and update testing protocols before regulatory mandates force reactive compliance.
| Substance Category | Testing Challenge | Terminology Impact |
|---|---|---|
| Synthetic opioids | Structural analogs evade standard panels | Requires specific fentanyl analog terminology |
| Designer stimulants | Novel compounds lack established cutoffs | Introduces provisional threshold language |
| Cannabis products | THC concentration variability in edibles and concentrates | Differentiates between THC and THCA terminology |
| Alcohol biomarkers | Ethyl glucuronide extends detection beyond breathalyzers | Adds EtG terminology to alcohol testing vocabulary |
Fentanyl’s rise fundamentally altered drug testing terminology by introducing nanogram-level sensitivity requirements and metabolite-specific language. You now encounter terms like norfentanyl, carfentanil, and acetylfentanyl in laboratory reports, each representing distinct compounds requiring separate detection capabilities. Synthetic cannabinoids and cathinones (bath salts) introduced entirely new drug classes absent from traditional five-panel tests, forcing terminology expansion to accommodate these threats.
Alcohol intoxication remains the leading cause of substance-related visits, yet workplace testing often focuses exclusively on illicit drugs. This discrepancy creates terminology gaps where organizations lack vocabulary for discussing alcohol biomarkers like phosphatidylethanol (PEth) or ethyl glucuronide (EtG) that detect consumption days after traditional breathalyzer windows close. Marijuana legalization in numerous states introduces terminology distinguishing between recreational use, medical use, and impairment, with no consensus on appropriate workplace policies.
Updating your terminology and protocols in response to evolving substance use requires systematic steps:
- Review epidemiological data annually to identify emerging substances affecting your workforce or service population, noting new drug names and street terminology.
- Assess whether current testing panels detect identified substances or if panel expansion is necessary, consulting laboratory partners about available assays and cutoff options.
- Update written policies to incorporate new terminology, ensuring consistency across employee handbooks, consent forms, and result notification letters.
- Train supervisors and medical review officers on new terminology, providing examples of how emerging substances appear in laboratory reports and what clinical significance they carry.
- Communicate changes to tested populations using clear, accessible language that explains why new substances entered testing protocols and what constitutes prohibited use.
Demographic trends affect testing terminology as substance use patterns vary by age, gender, occupation, and geographic region. Younger workers show higher rates of marijuana and synthetic drug use, while older workers demonstrate elevated prescription opioid misuse. Gender differences appear in benzodiazepine use rates and alcohol consumption patterns. These variations require you to understand terminology around demographic risk factors when designing targeted testing strategies or interpreting population-level results.
Implementing multi-panel drug testing guide strategies helps you address diverse substance use patterns with comprehensive detection capabilities. Expanded panels that include synthetic opioids, benzodiazepines, and barbiturates use terminology reflecting broader threat recognition. Understanding panel configuration terminology ensures your procurement specifications match your risk assessment and regulatory obligations.
Enhance your drug testing program with trusted supplies and expert guidance
Navigating drug testing terminology becomes significantly easier when you partner with suppliers who understand compliance requirements and provide clear product specifications. Buy Test Cup offers comprehensive drug test cups designed for government agencies, healthcare facilities, and corporate compliance programs requiring reliable results and regulatory alignment. Our product descriptions use precise terminology that maps directly to federal standards, eliminating guesswork about whether specific tests meet your obligations.
Beyond products, we provide educational resources that clarify complex terminology and guide implementation decisions. Our drug testing supplies checklist helps you identify all components needed for compliant testing programs, using terminology that connects supplies to specific workflow steps. Understanding how collection containers, temperature strips, adulterant checks, and chain of custody forms work together prevents gaps that compromise result validity.
For organizations building or refining testing programs, our drug testing program workflow resource explains how terminology applies at each stage from policy development through result reporting. This guidance helps you implement consistent processes that withstand audits and legal scrutiny while treating tested individuals fairly.
Frequently asked questions about drug testing terminology and compliance
What is the difference between screening and confirmatory drug tests?
Screening tests use immunoassays to rapidly detect drug classes above specified cutoff concentrations, providing presumptive results in minutes. Confirmatory tests employ mass spectrometry to verify screening results with high specificity, identifying exact compounds and concentrations for legal defensibility. Only confirmatory results should trigger adverse employment actions or legal consequences.
How do federal regulations affect drug testing panels in 2026?
Federal regulations at 42 CFR and 49 CFR define mandatory drug classes, cutoff concentrations, and testing procedures for covered employers. In 2026, these regulations include fentanyl and norfentanyl due to overdose prevalence, requiring panel updates and lower detection thresholds. Non-regulated employers may adopt these standards voluntarily or customize panels based on specific risks.
What accommodations do employers need to provide related to drug testing under the ADA?
Employers must engage in an interactive process to identify reasonable accommodations for applicants with disabilities affecting their ability to complete standard drug testing. Accommodations might include modified collection procedures, alternative specimen types, extended timeframes, or accessible testing locations. Documentation of this process using specific terminology protects against discrimination claims.
How has fentanyl inclusion changed drug testing terminology recently?
Fentanyl’s addition to regulated panels introduced terminology around nanogram-level sensitivity, metabolite detection (norfentanyl), and analog cross-reactivity. Testing protocols now distinguish between traditional opiates and synthetic opioids, requiring separate detection capabilities and result interpretation frameworks. This change reflects fentanyl’s role in the overdose crisis and its distinct pharmacology compared to heroin or prescription opioids.
What are common challenges interpreting drug test results?
Interpretation challenges include false positives from cross-reactive substances, false negatives when concentrations fall below cutoffs, variable detection windows based on individual metabolism, and clinical relevance questions about whether positive results indicate impairment or remote use. Medical review officers apply clinical judgment and interview donors to resolve these ambiguities before reporting final results.