Blog
Laboratory drug tests: 2026 workplace safety guide

Many workplace safety managers believe laboratory drug tests produce immediate results, yet specimens require metabolic processing before detection becomes possible. This misconception causes confusion when timing tests after incidents or accidents. Our guide breaks down test types, detection windows, specimen handling, and compliance requirements so you can make informed decisions that protect both employees and your organization from costly mistakes.
Key takeaways
| Point | Details |
|---|---|
| Testing combines speed and precision | Immunoassay screens flag potential positives quickly, while mass spectrometry confirms exact metabolites for definitive results. |
| Specimen choice affects detection | Urine detects days of use, saliva captures hours, hair reveals months, and blood shows current impairment. |
| Collection integrity prevents disputes | Chain-of-custody documentation and adulterant checks secure specimen validity and legal defensibility. |
| Regulatory compliance requires certified labs | SAMHSA certification, proficiency testing, and quality controls ensure trustworthy workplace drug testing. |
| Common errors undermine test trust | False positives, cutoff misreads, and timing errors create workplace disputes when not properly understood. |
Introduction to laboratory drug testing in workplaces
Laboratory drug tests analyze biological specimens to detect drugs and their metabolites using scientifically validated methods. These tests serve as crucial tools for maintaining safe work environments, protecting employee health, and meeting regulatory obligations across industries. Organizations deploy testing programs to identify substance use that could compromise safety, productivity, or compliance with federal and state regulations.
Workplace testing typically screens for specific drug classes that pose safety risks. Common target substances include:
- Opioids such as heroin, morphine, codeine, and prescription painkillers
- Cocaine and its metabolites
- Amphetamines including methamphetamine and prescription stimulants
- Tetrahydrocannabinol from marijuana products
- Phencyclidine and other hallucinogens
- Benzodiazepines used for anxiety treatment
- Barbiturates and other sedatives
Several workplace scenarios trigger testing requirements. Random screening programs deter ongoing substance use by selecting employees unpredictably throughout the year. Post-incident testing investigates whether impairment contributed to accidents, injuries, or equipment damage. Pre-employment screening verifies candidates meet safety standards before hire. Reasonable suspicion testing addresses observable behaviors suggesting impairment. Return-to-duty protocols confirm abstinence before reinstating employees after violations. For comprehensive workplace drug testing safety compliance strategies that protect your organization, understanding these testing contexts helps you implement programs that balance safety needs with employee rights.
Pro Tip: Document your testing policy clearly in employee handbooks and train supervisors on recognizing reasonable suspicion indicators to ensure consistent, defensible testing decisions.
Effective programs require detailed procedures covering every testing phase. The 7-step employee drug testing checklist provides a framework for establishing protocols that withstand legal scrutiny while maintaining workplace safety.
How laboratory drug tests work: from screening to confirmation
Laboratory drug tests predominantly use immunoassay screening followed by confirmatory tests such as GC-MS or LC-MS/MS for accurate identification. Immunoassay technology detects drug classes rapidly using antibody reactions that bind to target substances or similar compounds. This speed makes screening practical for high-volume workplace testing, delivering preliminary results within minutes to hours.
However, immunoassays have limitations. Cross-reactivity occurs when antibodies bind to substances structurally similar to target drugs, creating false positive results. Common medications, dietary supplements, or even poppy seeds can trigger positive screens without actual drug use. This is why presumptive positive results always require confirmation.
Confirmatory testing employs gas chromatography-mass spectrometry or liquid chromatography-tandem mass spectrometry to identify specific drug metabolites with near-absolute certainty. These methods separate specimen components, then measure precise molecular weights to confirm exact substances present. Unlike immunoassays, mass spectrometry eliminates false positives by detecting unique metabolite signatures.
Understanding parent drugs versus metabolites clarifies what tests actually measure. Parent drugs are the original substances consumed, while metabolites are breakdown products your body creates during drug processing. Tests primarily detect metabolites because they persist longer in specimens and provide more reliable evidence of use. For instance, THC metabolizes into THC-COOH, which remains detectable in urine days after the psychoactive effects fade.
| Testing Method | Speed | Cost | Accuracy | Result Type |
|---|---|---|---|---|
| Immunoassay screening | Minutes to hours | Low | Moderate (prone to cross-reactivity) | Presumptive positive or negative |
| GC-MS confirmation | Days | High | Very high (identifies specific metabolites) | Definitive positive or negative |
| LC-MS/MS confirmation | Days | High | Very high (detects multiple compounds simultaneously) | Definitive positive or negative |
Pro Tip: Never take adverse employment action based solely on immunoassay results. Always wait for mass spectrometry confirmation to avoid wrongful termination claims from false positives.
For diverse workplace needs, reviewing examples of workplace drug tests for HR managers helps you select appropriate testing panels matching your industry risks and regulatory requirements.
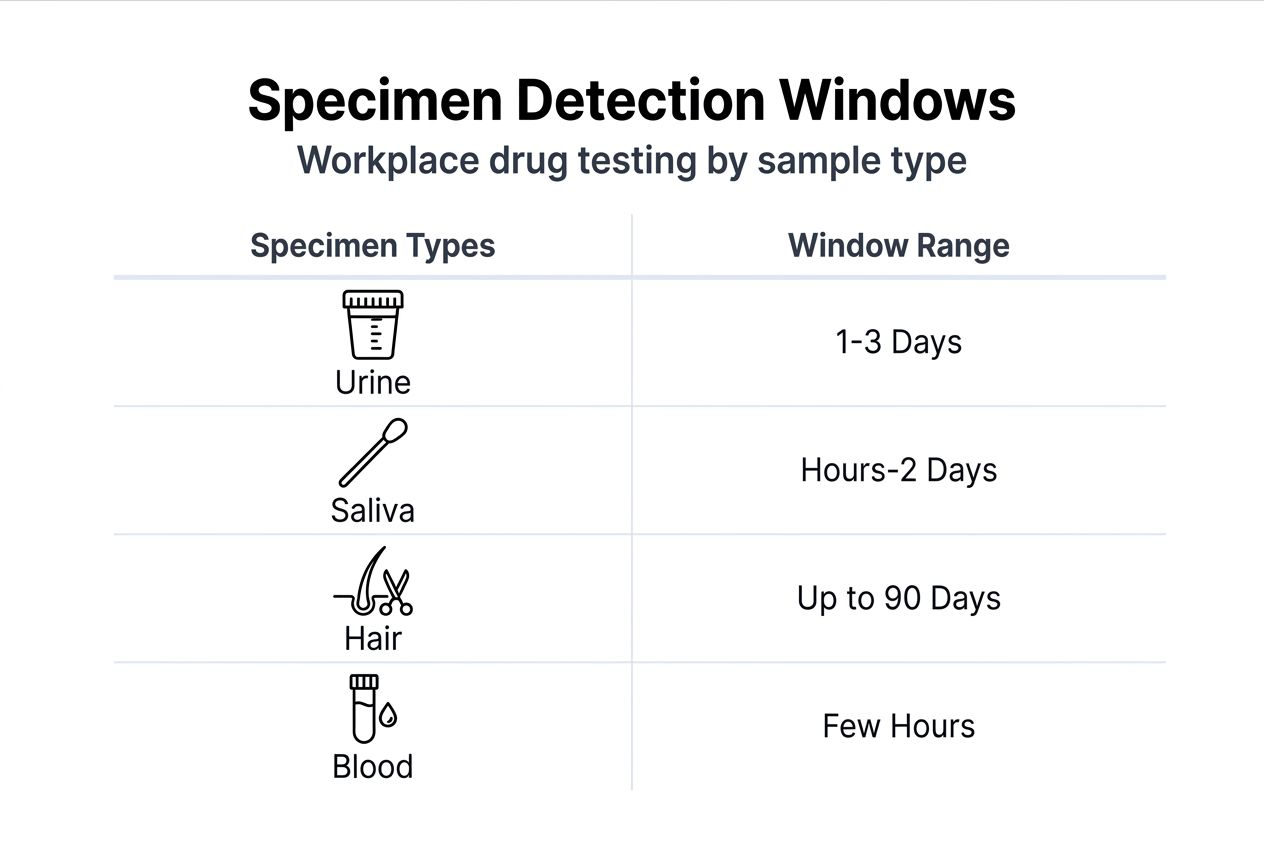
Specimen types and detection windows in workplace testing
Detection windows vary significantly by specimen type, directly affecting which test format suits your workplace needs. Urine remains the most common specimen because it balances practicality with a detection window spanning one to four days for most drugs. Collection is noninvasive, costs stay reasonable, and established cutoff standards support consistent interpretation.
Saliva testing captures recent use within hours to two days, making it ideal for post-incident or reasonable suspicion scenarios where current impairment matters most. Collection requires simple oral swabs without privacy concerns, reducing collection site requirements and employee discomfort. However, shorter detection windows mean saliva misses use beyond the immediate timeframe.
Hair follicle testing extends detection up to 90 days, revealing patterns of repeated use rather than single incidents. This specimen type resists tampering and provides historical context, but hair grows slowly so recent use within the past week may not appear. Additionally, cosmetic treatments can reduce detectable metabolite levels, complicating interpretation.
Blood testing offers the shortest window, detecting active drug presence for mere hours. This precision identifies current impairment but requires invasive collection by trained phlebotomists, limiting practical workplace applications. Blood tests typically serve post-accident investigations where immediate impairment determination outweighs cost and logistics concerns.
| Specimen Type | Detection Window | Collection Method | Tampering Risk | Best Use Case |
|---|---|---|---|---|
| Urine | 1 to 4 days (longer for chronic use) | Observed or unobserved void | Moderate (dilution, substitution) | General workplace screening |
| Saliva | Hours to 2 days | Oral swab | Low (difficult to adulterate) | Post-incident, reasonable suspicion |
| Hair | Up to 90 days | Cut close to scalp | Very low | Pattern detection, pre-employment |
| Blood | Hours | Venipuncture | Very low (medical setting) | Accident investigation |
Pro Tip: Match specimen type to your testing purpose. Use saliva for immediate impairment questions, urine for routine screening, and hair when you need historical use patterns for safety-sensitive positions.
Balancing detection needs against practical constraints requires understanding each specimen’s strengths. The workplace drug screening overview explores how different industries select specimen types based on regulatory requirements and operational realities.
Sample collection and handling best practices
Chain-of-custody procedures prevent specimen tampering and contamination from collection through laboratory analysis. Every person handling specimens must document transfers using standardized forms recording dates, times, and signatures. This paper trail proves specimen integrity if results face legal challenges during disputes or litigation.
Adulteration detection testing identifies attempts to tamper with urine specimens. Common adulterants include bleach, vinegar, commercial masking products, and excessive water to dilute drug concentrations below detection cutoffs. Testing specimen pH, specific gravity, creatinine levels, and temperature immediately after collection reveals manipulation attempts. Specimens outside normal physiological ranges trigger closer scrutiny or recollection.
Proper collection procedures minimize errors:
- Verify donor identity using photo identification before collection begins
- Secure the collection area removing potential contaminants or adulterants
- Instruct donors on proper technique without contaminating the specimen
- Observe or monitor collection when policy and regulations require direct observation
- Measure specimen temperature within four minutes to confirm fresh provision
- Test specimen validity markers including pH and specific gravity immediately
- Seal specimens in tamper-evident containers with donor and collector signatures
- Complete chain-of-custody documentation without leaving gaps in the timeline
- Store specimens at proper temperatures until laboratory transport occurs
- Ship specimens to certified laboratories using approved courier services
Pro Tip: Train collection site staff annually on procedures and adulterant recognition. Regular refreshers prevent complacency that creates vulnerabilities in your testing program.
Documentation standards protect organizations when employees challenge results. Incomplete forms, signature gaps, or temperature recording failures can invalidate otherwise accurate tests. For systematic approaches, the drug testing program workflow compliance guide details documentation requirements that withstand audit scrutiny.
Common misconceptions and errors in laboratory drug testing
Delay between drug consumption and laboratory detectability confuses many workplace decision makers. Drugs require time for absorption, distribution, metabolism, and excretion before metabolites accumulate in specimens at detectable concentrations. Testing immediately after suspected use often produces negative results despite actual consumption, leading to false confidence about employee sobriety.
False positives in immunoassay screening stem from antibody cross-reactivity with structurally similar compounds. Over-the-counter medications, prescription drugs, and even some foods trigger positive screens without illicit drug use. Common culprits include:
- Ibuprofen causing false positives for marijuana
- Decongestants triggering amphetamine alerts
- Quinolone antibiotics flagging as opioids
- Poppy seed consumption showing opiate presence
Confirmatory testing resolves these issues by identifying exact metabolites, eliminating false accusations and wrongful terminations. Organizations taking action on screening results alone face legal liability and damaged employee relations.
Cutoff levels represent concentration thresholds separating positive from negative results. Values below cutoffs receive negative interpretations even when trace amounts exist, preventing positive calls from passive exposure or distant use. Misunderstanding cutoffs leads some to believe any detectable amount constitutes a positive, but standardized cutoffs balance sensitivity against specificity to ensure meaningful workplace decisions.
Hair testing limitations include cosmetic interference and growth delay gaps. Bleaching, dyeing, and chemical straightening reduce metabolite concentrations, potentially converting true positives into false negatives. Additionally, hair requires time to grow beyond the scalp before collection, creating a window where very recent use escapes detection.
Immunoassay false positive rates range from 5% to 10% depending on the drug class tested, making confirmatory mass spectrometry essential for accurate workplace decisions.
Addressing these misconceptions through education prevents testing program failures. The employee drug testing checklist incorporates safeguards against common errors that undermine program credibility.
Regulatory compliance and quality standards for drug testing laboratories
SAMHSA certification establishes federal standards for workplace drug testing laboratories. The Substance Abuse and Mental Health Services Administration mandates strict quality controls, personnel qualifications, and proficiency testing for labs analyzing federal workforce specimens. While not all private employers require SAMHSA certification, choosing certified labs ensures testing meets the highest reliability standards.
Proficiency testing validates laboratory accuracy through blind samples with known drug concentrations. External agencies send specimens quarterly, and labs must correctly identify substances and concentrations to maintain certification. Consistent proficiency failures trigger investigations, corrective actions, or certification revocation, protecting you from unreliable results.
Laboratory accreditation requirements include:
- Personnel holding appropriate scientific credentials and training
- Validated testing procedures following manufacturer specifications
- Regular equipment calibration and maintenance documentation
- Quality control samples run with each testing batch
- Comprehensive standard operating procedure manuals
- Secure specimen storage preventing degradation or mix-ups
- Participation in external proficiency testing programs
- Internal audits verifying procedure adherence
Standardized reporting practices ensure consistent result interpretation across laboratories. Medical review officers receive detailed reports showing specimen validity, screening results, confirmatory findings, and specific metabolite concentrations. This standardization prevents confusion when interpreting results from different testing facilities.
For programs requiring random selection protocols, understanding random drug testing compliance in 2026 helps maintain defensible selection processes. Comparing methodologies through a drug testing methods comparison guide ensures your chosen approach aligns with current regulatory expectations.
Interpretation and reporting of drug test results
Interpreting laboratory results requires understanding metabolite significance and cutoff applications. Confirmatory tests report exact metabolite concentrations in nanograms per milliliter. Values exceeding established cutoffs receive positive interpretations, while lower concentrations get reported as negative despite detectable presence. This framework prevents positive calls from insignificant exposures or laboratory background noise.
Borderline results near cutoff thresholds demand careful handling. Some laboratories retest specimens approaching cutoffs to confirm concentrations before final reporting. Medical review officers evaluate borderline findings considering specimen validity, testing limitations, and donor explanations. Rushing judgment on marginal results risks errors that damage employee trust and expose organizations to disputes.
Legal and ethical result communication principles protect both employers and employees:
- Maintain strict confidentiality limiting access to need-to-know personnel
- Report results only to designated management representatives
- Provide employees opportunities to explain positive findings
- Document all result communications with dates and recipients
- Avoid discussing results in settings where others might overhear
- Follow consistent procedures for all employees regardless of position
- Retain records according to federal and state requirements
Pro Tip: Establish standardized result reporting templates specifying who receives notifications, required response timeframes, and documentation standards. Consistency prevents discrimination claims and ensures fair treatment.
Medical review officers serve as physician intermediaries reviewing positive results before employer notification. They interview donors about medications, medical conditions, or other legitimate explanations for positive findings. This step protects employees with valid prescriptions from wrongful adverse actions while confirming true policy violations.
For organizations implementing comprehensive panels, the guide to multi-panel drug testing explains how to interpret results across multiple substance categories simultaneously.
Conclusion: best practices for reliable laboratory drug testing in workplaces
Selecting appropriate testing methods requires balancing detection windows against accuracy needs and practical constraints. Match specimen types to your specific workplace scenarios, using urine for general screening, saliva for immediate impairment questions, and hair when historical patterns matter. Prioritize confirmatory testing over screening-only approaches to eliminate false positives that damage employee relations.
Strict specimen collection protocols protect test integrity:
- Implement comprehensive chain-of-custody procedures without documentation gaps
- Deploy adulterant testing catching dilution and substitution attempts
- Train collection site personnel on proper techniques and tampering recognition
- Maintain temperature and validity checks for every specimen
Regulatory compliance through certified laboratory partnerships ensures defensible results. Choose SAMHSA-certified facilities when federal standards apply, or select accredited labs meeting equivalent quality benchmarks for private sector testing. Verify laboratories participate in proficiency testing and maintain current certifications.
Avoid interpretation errors by understanding cutoff significance, metabolite detection principles, and false positive causes. Never take adverse action on preliminary screening results alone. Engage medical review officers to evaluate positive confirmations, considering legitimate medical explanations before assuming policy violations. Document every testing decision thoroughly to withstand potential legal challenges.
Enhance your workplace drug testing program with trusted supplies
Maintaining specimen integrity starts with quality collection supplies designed for workplace testing demands. Reliable drug test cups with built-in adulterant checks and temperature strips help you detect tampering attempts immediately. Accurate drug test strips provide quick preliminary results while you await laboratory confirmation.
Our certified products meet regulatory standards, ensuring your testing program delivers trustworthy results that support compliance goals. We serve government agencies, workplaces, and organizations requiring bulk testing solutions with same-day shipping options. Explore our comprehensive catalog and drug testing program workflow compliance resources to implement best practices that protect your workforce.
Frequently asked questions
What factors influence the detection window in laboratory drug testing?
Detection windows depend primarily on specimen type and individual metabolism rates. Urine typically captures use from one to four days, saliva detects recent hours to two days, hair reveals up to 90 days of history, and blood shows only current hours of presence. Chronic users show longer detection periods than occasional users due to drug accumulation in body tissues.
How do false positives occur in immunoassay screening, and how are they resolved?
False positives arise when screening antibodies bind to non-target substances structurally similar to tested drugs. Common medications like ibuprofen, decongestants, and certain antibiotics trigger positive screens without actual drug use. Confirmatory testing via gas chromatography-mass spectrometry or liquid chromatography-tandem mass spectrometry identifies exact metabolites, eliminating false positives and providing definitive results.
What is the importance of chain-of-custody in sample collection?
Chain-of-custody documentation tracks every person handling specimens from collection through laboratory analysis and result reporting. This paper trail prevents tampering, contamination, or specimen mix-ups that could invalidate results. Proper chain-of-custody ensures test validity and provides legal defensibility when employees challenge findings in disputes or litigation.
Can cosmetic hair treatments affect hair drug test results?
Hair treatments including bleaching, dyeing, perming, and chemical straightening can reduce detectable drug metabolite concentrations significantly. These cosmetic processes damage hair structure and leach metabolites, potentially converting true positives into false negatives. Results should be interpreted carefully when donors report recent cosmetic treatments, possibly requiring alternative specimen types for accurate assessment.