Blog
Interpreting Drug Test Results: Accuracy & Compliance

TL;DR:
- Proper interpretation and confirmatory testing are essential to avoid legal and compliance risks.
- Specimen type determines detection window and suitability for investigative goals.
- Following procedural protocols and using certified labs ensure result accuracy and legal defensibility.
Misreading a drug test result is not a minor administrative slip. It can trigger wrongful termination claims, regulatory sanctions, or a failed audit that puts your entire program at risk. False positive rates between 5-10% from initial immunoassay screens mean that a significant share of presumptive positives in any program are not confirmed drug use. For compliance officers, clinic managers, and workplace administrators, the gap between a raw result and a defensible conclusion is where most errors happen. This guide walks through specimen types, cutoff thresholds, result validity, and confirmation protocols so you can interpret results with confidence and stay on the right side of federal standards.
Key Takeaways
| Point | Details |
|---|---|
| Specimen matters | Urine, oral fluid, and hair offer different detection windows and compliance contexts. |
| Understand cutoffs | Drug test cutoff levels determine if results are considered negative or non-negative for compliance. |
| Verify with confirmation | Always require laboratory confirmation of non-negatives to avoid false positives and ensure defensibility. |
| MRO critical role | An MRO ensures non-negatives are professionally reviewed, including medical and prescription history. |
| Lab accuracy is key | Relying on certified labs and modern confirmation methods prevents costly interpretation errors. |
Understanding drug test types and detection windows
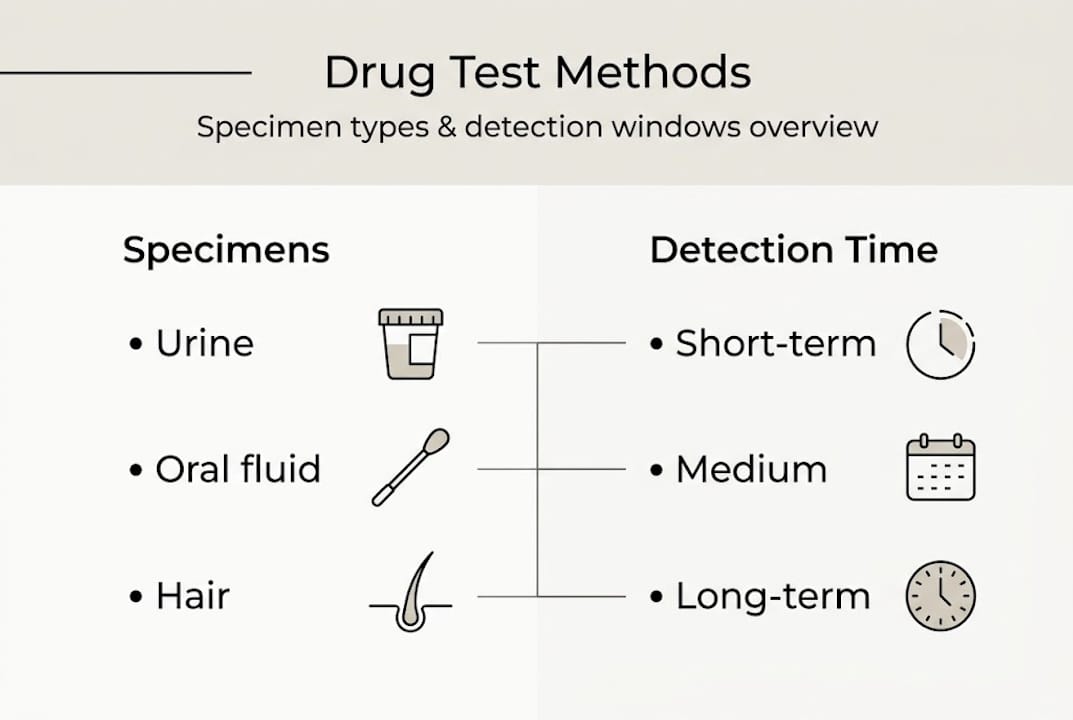
Let’s start by clarifying how different specimen types influence what a result actually means. The specimen you collect determines the detection window, the applicable cutoffs, and ultimately what period of drug use the result reflects. Choosing the wrong specimen for your investigative purpose is one of the most underappreciated errors in compliance programs.
Specimen types each have distinct cutoffs and detection windows that directly shape how you interpret any result. Urine is the most widely used specimen and detects drug metabolites for roughly 1 to 4 days after use for most substances, though chronic heavy use can extend that window significantly. Oral fluid tests are less invasive and better suited for detecting very recent use, with windows typically up to 48 hours. Hair testing windows reach up to 90 days, making it the preferred option when long-term use history matters more than recent exposure.
Here is a quick comparison to keep your selection decisions grounded:
| Specimen type | Detection window | Invasiveness | Court acceptance | Best use case |
|---|---|---|---|---|
| Urine | 1 to 4 days (typical) | Low | High | Standard workplace, DOT |
| Oral fluid | Up to 48 hours | Very low | Moderate | Post-accident, recent use |
| Hair | Up to 90 days | Low | Growing | Long-term history, pre-employment |
Key considerations when selecting a specimen type:
- Urine requires specimen validity testing to detect dilution, substitution, or adulteration
- Oral fluid is harder to adulterate but has narrower detection windows and lower cutoffs
- Hair cannot detect very recent use (within the last 7 days) due to growth lag
- Federal regulated programs (DOT, HHS) have specific approved specimen types per program
For a deeper look at how these options stack up in practice, you can compare drug testing methods across compliance scenarios.
Pro Tip: Match your specimen type to your investigative goal. If you need to know whether someone used drugs in the last 24 hours, oral fluid is your tool. If you need a 90-day picture for pre-employment screening, hair is the right call.
Cutoff levels and federal compliance standards
Once you’ve selected a specimen type, the next key to correct interpretation is understanding cutoff levels and regulatory context. A cutoff is not a measure of impairment. It is a legal and technical threshold. Results below the cutoff are reported as negative regardless of whether a trace amount of a substance is present. Results at or above the cutoff are non-negative and trigger the next step in your protocol.
SAMHSA sets mandatory cutoffs for key drug classes, and as of 2026, expanded panels now include fentanyl, reflecting the ongoing public health response to the opioid crisis. These cutoffs apply to federal workplace drug testing programs and serve as the benchmark for most regulated industries.
Current screening and confirmation cutoffs for core drug classes:
| Drug class | Screen cutoff (urine) | Confirmation cutoff (urine) | Oral fluid screen cutoff |
|---|---|---|---|
| THC (marijuana) | 50 ng/mL | 15 ng/mL | 4 ng/mL |
| Cocaine metabolites | 150 ng/mL | 100 ng/mL | 8 ng/mL |
| Opiates (codeine/morphine) | 2,000 ng/mL | 2,000 ng/mL | 40 ng/mL |
| Amphetamines | 500 ng/mL | 250 ng/mL | 50 ng/mL |
| PCP | 25 ng/mL | 25 ng/mL | 10 ng/mL |
| Fentanyl (expanded panel) | 0.5 ng/mL | 0.2 ng/mL | Varies |
Key points about cutoff application:
- Cutoffs are not impairment thresholds. A result below 50 ng/mL for THC is reported negative even if metabolites are detectable
- Confirmatory cutoffs are often lower than screening cutoffs, which is intentional to reduce false negatives at the confirmation stage
- Non-federal employers may adopt federal cutoff standards voluntarily or set their own thresholds within legal limits
- Understanding the difference between a screen result and a confirmed result is foundational to correct interpretation; review drug screening terms if your team needs a refresher
What influences positive, negative, and invalid results?
Understanding what a result means also demands context about false positives, negatives, and the crucial review process. A positive on an initial screen is not a conclusion. It is a flag that requires follow-up.
Immunoassays are prone to 5 to 10% false positive rates, and prescription medicines can trigger positives that have nothing to do with illicit drug use. Common cross-reactivity triggers include ibuprofen producing a false positive for THC, pseudoephedrine (found in many cold medicines) triggering amphetamine positives, and poppy seeds causing opiate positives. These are not edge cases. They happen regularly in active testing programs.
“5 to 10% of initial immunoassay screens may be false positives, requiring confirmatory testing before any adverse action.”
False negatives are equally important to understand. A negative result does not always mean the person is drug-free. Diluted specimens, drug concentrations just below the cutoff, testing outside the detection window, or deliberate adulteration can all produce a negative result that does not reflect reality.
Invalid results occur when specimen validity testing detects tampering, substitution, or a sample that cannot be analyzed. These require a separate protocol response, not a simple re-test without investigation.
Steps for handling a non-negative result:
- Flag the result as non-negative and document the chain of custody
- Send the specimen to an HHS-certified laboratory for confirmation testing
- Refer the confirmed non-negative to a certified Medical Review Officer for review
- The MRO contacts the donor to assess legitimate prescription use or medical explanation
- The MRO issues a final verified result (positive, negative, or canceled)
- HR or compliance acts only on the MRO-verified final result
Pro Tip: Never take a non-negative result directly to HR or management. The MRO step is not optional in regulated programs. Skipping it is a compliance violation, not just a procedural shortcut. For broader context, the workplace safety guide covers how this fits into a complete program, and ensuring accurate screening outlines the technical safeguards that reduce error rates.
Confirmation testing and laboratory accuracy
When a non-negative appears, confirmation and accuracy become top priorities. The screening test got you a flag. The confirmation test gets you the truth.
Immunoassays are screens; confirmation tests are legally required for non-negatives and resolve most immunoassay errors. The confirmation method of choice is LC-MS/MS, which stands for liquid chromatography tandem mass spectrometry. It identifies specific drug molecules rather than reacting to antibody cross-reactivity, which is why it eliminates the false positive problem almost entirely.
LC-MS/MS is the gold standard for legally defensible confirmations and is required for all federal workplace drug testing programs. No other method provides the same specificity and sensitivity at the confirmation stage.
The pathway from screening to confirmed result:
- Step 1: Point-of-care or laboratory immunoassay screen produces a presumptive result
- Step 2: Non-negative specimens are transferred to an HHS-certified lab under documented chain of custody
- Step 3: LC-MS/MS confirmation is performed on the same specimen
- Step 4: Confirmed result is reported to the MRO, not directly to the employer
- Step 5: MRO review and verification produces the final reportable result
Laboratory selection is not a minor procurement decision. Only HHS-certified laboratories are authorized to perform confirmation testing for federally regulated programs. Using a non-certified lab invalidates the result for compliance purposes. For more on this, the lab accuracy importance resource covers what to look for when evaluating lab partners, and accurate drug screening ties the full process together.
Pro Tip: A presumptive positive from a point-of-care cup is a starting point, not a finding. No employment action, policy consequence, or legal referral should occur until LC-MS/MS confirmation and MRO review are complete.
A fresh perspective: Common pitfalls in interpreting drug test data
With the technical framework in mind, here’s what most guides miss about the real-world pitfalls of interpreting drug test results. The biggest mistake we see in compliance programs is treating the result as the conclusion rather than the beginning of a process. A line on a test cup, a number on a lab report, a flag in a software system — none of these are decisions. They are data points.
The programs that get into legal trouble are almost never the ones that missed a positive. They are the ones that acted on an unverified result, skipped the MRO step, or failed to document chain of custody. Rigid interpretation without procedural context is not compliance. It is liability.
Training matters more than most administrators realize. The person collecting the specimen, the person logging the result, and the person communicating it to HR all need to understand their role in a chain, not just their individual task. One break in that chain can make an otherwise clean result legally indefensible.
Familiarizing your team with professional testing terminology is a practical first step that most programs overlook until something goes wrong.
Pro Tip: Treat every non-negative as a process, never a conclusion. The result initiates the protocol. The protocol produces the decision.
Get reliable tools and resources for compliant drug testing
Accurate interpretation depends on starting with trusted, compliant tools — the right resources make all the difference. Every step in your testing protocol, from collection to result, depends on the quality and compliance of the supplies you use. Substandard cups, improperly stored strips, or non-validated collection kits introduce error before a single result is read.
At Buy Test Cup, we supply government agencies, clinics, and compliance-focused workplaces with drug test cups, dip strips, oral swabs, and specimen collection kits built for accuracy and regulatory alignment. If you’re building or auditing your program, start with the essential drug testing supplies checklist to make sure nothing critical is missing. For end-to-end process support, the drug screening workflow guide walks through each stage from collection to final result.
Frequently asked questions
Who verifies and interprets non-negative drug test results?
A certified Medical Review Officer reviews all non-negative results, checking the donor’s medical history and any legitimate prescriptions before issuing a final verified report to the employer.
Why are confirmation tests required after a positive screening?
LC-MS/MS confirmation eliminates false positives produced by immunoassay cross-reactivity, providing the specificity and legal defensibility required for any adverse action under federal compliance programs.
What are the most common reasons for false negatives?
Diluted samples, drug concentrations just below the cutoff, testing outside the substance’s detection window, or deliberate specimen adulteration can all produce a negative result that does not reflect actual drug use.
What specimen types are accepted for federally regulated drug testing?
Urine remains the primary standard, but oral fluid and hair are permitted under specific agency guidelines, each carrying distinct detection windows, cutoff values, and chain-of-custody requirements.
How are adulterated or invalid results handled?
Specimen validity tests identify tampering indicators; the MRO then reviews the test integrity findings and may require a directly observed retest or cancel the result per federal protocol.