Blog
Step-by-step urine test procedure for accurate compliance

A single procedural misstep during urine drug testing can unravel months of compliance work, expose your organization to legal liability, and undermine the trust of every stakeholder involved. Whether you manage testing programs for a federal agency, a hospital network, or a corporate fleet, the margin for error is essentially zero. SAMHSA guidelines and DOT 49 CFR Part 40 set the standard, and this guide walks you through every phase of the process, from site setup to chain of custody, so your program holds up under any audit or legal review.
Key Takeaways
| Point | Details |
|---|---|
| Follow detailed protocol | Strictly adhering to each step ensures tests are legally defensible and reliable. |
| Verify sample integrity | Essential checks like temperature, volume, and validity protect against costly errors or disputes. |
| Chain of custody matters | Documented handoffs using the CCF safeguard compliance from collection to lab. |
| Understand testing methods | Knowing screening versus confirmation steps prevents misinterpretation and failed audits. |
| Handle exceptions correctly | Special protocols for shy bladder, observed collections, and tampering keep results valid. |
Preparation: What you need before collection
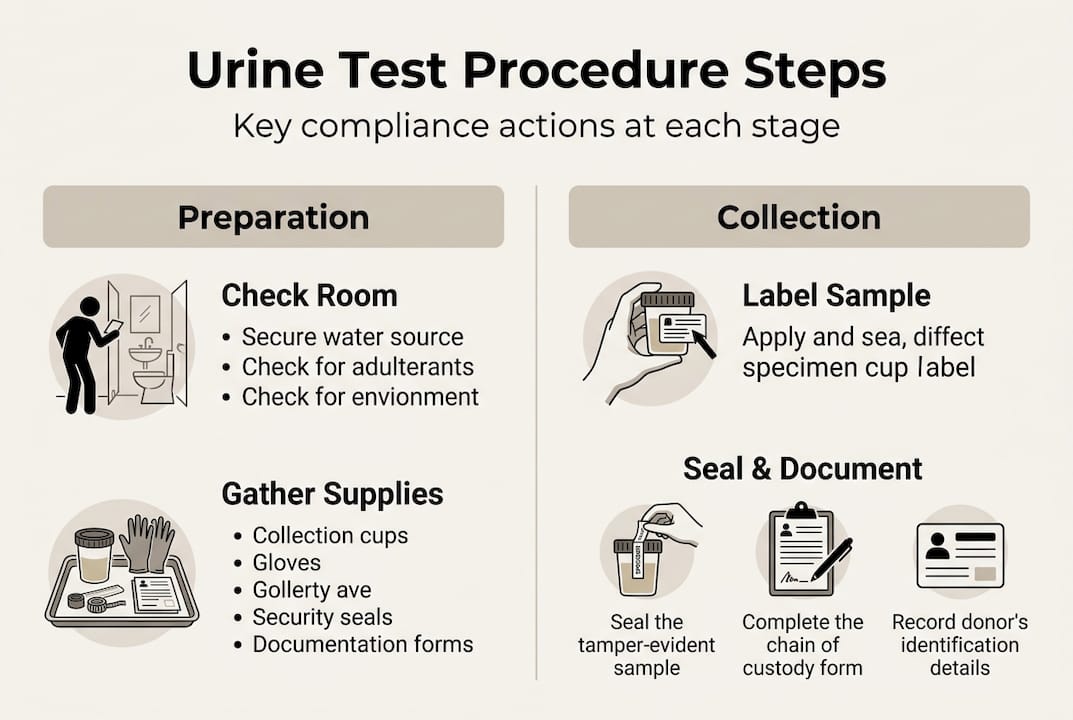
Before a single donor walks through the door, your collection site must meet specific environmental and administrative requirements. Skipping this groundwork is one of the most common reasons programs fail audits. Think of preparation as your legal foundation: everything built on top of it depends on how solid it is.
The SAMHSA collection requirements are clear: you need a single-toilet restroom with blue dye added to the toilet water, tamper-evident collection kits, and secure storage for donor belongings. The room must be free of any water sources a donor could use to dilute or substitute a specimen. Access to federal workplace drug testing standards will help you verify your site meets every physical requirement.
Here is a quick-reference checklist for your pre-collection setup:
- Environment: Single-toilet restroom, blue dye in toilet, no accessible water sources, blued or taped faucets
- Supplies: Tamper-evident collection cups, Federal Chain of Custody Forms (CCF), specimen transport bags, temperature strips
- Documents: Donor ID verification log, employer authorization form, completed CCF header information
- Accessibility: ADA-compliant facilities, gender-appropriate observed collection staff if needed
- Security: Locked storage for donor personal items, restricted access to collection area
| Requirement | Federal standard | Why it matters |
|---|---|---|
| Toilet water color | Blue dye required | Detects substitution attempts |
| Collection cup | Tamper-evident, graduated | Ensures volume accuracy |
| CCF form | Federal 5-part form | Legal chain of custody |
| ID verification | Government-issued photo ID | Confirms donor identity |
| Storage | Locked, secure | Prevents tampering |
For a deeper look at urine drug testing guidance and compliance best practices, those resources can help you build a site-ready checklist tailored to your program.
Pro Tip: Pre-populate the non-sensitive sections of the CCF (employer name, address, MRO contact) before the donor arrives. This reduces errors under time pressure and keeps the collection moving efficiently.
Step-by-step urine test procedure: How to ensure accuracy
Once your setting and documents are ready, the main procedure unfolds in a repeatable, policy-aligned order. Every step below corresponds to federal requirements, and deviating from any one of them can compromise the legal defensibility of your results.
- Initiate the CCF. Record the specimen ID number, collection site, and employer information before the donor touches anything.
- Verify donor identity. Check a government-issued photo ID. Document the ID type and number on the CCF. No ID means no collection.
- Explain the process. Walk the donor through what will happen, including the temperature check and split specimen protocol. Keep it factual and neutral.
- Pre-collection steps. Instruct the donor to remove outer garments, empty pockets, and wash hands. Secure all personal belongings.
- Provide the collection container. Hand the donor a sealed, tamper-evident cup. Direct them to the restroom and explain the minimum volume requirement.
- Collection. The federal urine test workflow requires a minimum of 45mL total, with 30mL for the primary specimen (Bottle A) and 15mL for the split specimen (Bottle B).
- Temperature check. Read the temperature strip within 4 minutes of collection. The acceptable range is 90 to 100°F. An out-of-range reading triggers immediate action.
- Inspect the specimen. Note color and any unusual characteristics. Document everything on the CCF.
- Split and label. Pour and seal Bottle A and Bottle B. Apply tamper-evident labels. Have the donor initial both labels.
- Complete the CCF. Collector and donor both sign. Provide the donor with their copy.
- Package for shipment. Place both bottles in the transport bag with the lab copy of the CCF. Seal and ship to a certified laboratory.
For a full compliance drug test workflow reference, that resource covers edge cases and policy variations across industries.
| Scenario | Standard collection | Observed collection |
|---|---|---|
| Normal test | Donor collects privately | Not applicable |
| Temp out of range | Immediate second collection | Direct observation required |
| Tampering suspected | Document and escalate | Direct observation mandated |
| Prior refusal on record | Standard unless flagged | Observation may be required |
For a side-by-side look at testing methods comparison across collection types, that guide is worth reviewing before you finalize your program protocols.
Pro Tip: Document every irregularity in writing, no matter how minor it seems. A donor who refuses to initial the specimen label, walks away, or refuses to provide a sample is treated as a refusal to test under federal rules. That distinction matters enormously in legal proceedings.
Specimen validity checks and common pitfalls
Even with a complete collection, not every sample is valid. Validity testing is the quality control layer that separates a defensible result from a contested one.
Laboratories run a standard battery of validity checks on every specimen: creatinine levels must exceed 20 mg/dL, temperature must fall between 90 and 100°F at collection, and oxidant and adulterant screens must come back clean. Specific gravity and pH are also measured to detect dilution or substitution.
Here is what each result means for your program:
- Dilute specimen: Creatinine 2 to 20 mg/dL and specific gravity 1.001 to 1.003. May require a recollection under direct observation.
- Substituted specimen: Creatinine below 2 mg/dL and specific gravity outside 1.001 to 1.020. Treated as a refusal to test.
- Adulterated specimen: Presence of oxidants, surfactants, or other foreign substances. Treated as a refusal to test.
- Invalid result: Interfering substance detected but not identified. Requires a second collection, possibly observed.
- Out-of-range temperature: Below 90°F or above 100°F at the 4-minute mark. Requires immediate second collection under direct observation.
For a practical employee testing checklist that incorporates validity steps, that resource can help your HR team stay audit-ready.
For shy bladder situations, the SAMHSA FAQ on specimen validity is clear: donors have up to 3 hours to provide a sufficient specimen. After that, a medical evaluation is required. If no medical explanation exists, it is a refusal.
“Refusal to provide a specimen or tampering with a specimen equates to a refusal to test under federal rules, carrying the same consequences as a verified positive result.”
Testing methodologies: Screen and confirmation explained
Once the specimen is ready for lab testing, screening and confirmation methods protect you from false results. Understanding both is essential for anyone managing a compliant program.
Initial screening uses immunoassay technology, methods like EMIT, which are fast and cost-effective, typically running between $1 and $5 per test. Confirmation uses gas chromatography/mass spectrometry (GC/MS), which is highly specific and costs between $25 and $100 per test. A positive screen alone is never sufficient for a verified positive result. Confirmation is mandatory.
| Drug | Screening cutoff | Confirmatory cutoff |
|---|---|---|
| THC metabolite | 50 ng/mL | 15 ng/mL |
| Cocaine metabolite | 150 ng/mL | 100 ng/mL |
| Opiates | 2000 ng/mL | 2000 ng/mL |
| Amphetamines | 500 ng/mL | 250 ng/mL |
| Phencyclidine (PCP) | 25 ng/mL | 25 ng/mL |
These federal cutoff values are set by HHS and updated periodically. Always verify you are using the current year’s published limits.
Key points about the confirmation process:
- A Medical Review Officer (MRO) reviews all confirmed positives before results are reported to the employer
- The MRO contacts the donor to rule out legitimate medical explanations
- Only after MRO review is a result officially reported as positive or negative
- Donors may request Bottle B testing at a second certified lab if they dispute the result
For a detailed breakdown of drug testing methods and how they apply to your specific program, that guide covers both point-of-care and lab-based options. The workplace drug testing standards from NCBI also provide useful context on method selection.
Chain of custody: Ensuring defensibility from collection to reporting
Finalizing the process is not just about shipping a sample. The chain of custody protects your results in legal and compliance reviews, and any gap in documentation can invalidate an otherwise perfect collection.
The CCF tracks custody through five clear steps, each requiring signatures and timestamps:
- Donor step: Donor provides specimen, initials tamper-evident labels, and signs the CCF certifying the specimen is theirs.
- Collector step: Collector documents collection details, temperature, and any irregularities. Signs and dates the CCF.
- Employer/shipping step: Sealed specimens and CCF copies are packaged and shipped to the certified lab. Shipping manifest is retained.
- Laboratory step: Lab logs receipt, checks seal integrity, and assigns internal tracking. Any broken seal is documented and reported.
- MRO step: MRO receives lab results, reviews for medical legitimacy, and issues the final verified result to the employer.
Split specimens play a critical role here. Bottle A is the primary test specimen. Bottle B is stored frozen at the lab and only opened if the donor formally requests a retest through the MRO. This dual-bottle system is what makes federal results legally defensible.
For organizations building or updating their compliance drug testing policies, the federal chain of custody documentation requirements are a non-negotiable starting point.
Enhance your compliance and accuracy with trusted solutions
With the full procedure mastered, the next step is making sure your supplies and workflow tools match the standard you are working to uphold. At Buy Test Cup, we stock a full range of drug test cups that are tamper-evident, CLIA waived, and built for federal compliance. Whether you are running a high-volume government program or a clinic-based monitoring protocol, our catalog covers multi-panel cups, adulterant testing kits, and specimen collection devices. Use our workflow guide to align your internal process with current federal standards, and check the essential supplies for testing checklist to make sure nothing is missing before your next collection cycle. Bulk orders ship free, and same-day shipping is available for urgent program needs.
Frequently asked questions
What is the minimum urine sample volume for a federal urine test?
The required minimum is 45mL, split as 30mL for the primary specimen (Bottle A) and 15mL for the split specimen (Bottle B). Anything below this threshold requires a recollection.
When is direct observation required during a urine drug test?
Direct observation is mandated when tampering is suspected, the specimen temperature falls outside the 90 to 100°F range, or the donor has a prior refusal on record. It is not the default collection method.
How long are drugs typically detectable in urine?
Most drugs are detectable for 2 to 4 days after use in urine specimens. Chronic or heavy use can extend detection windows significantly beyond that range.
What happens if the donor cannot urinate on test day?
Donors experiencing shy bladder are given up to 3 hours to provide a sufficient specimen. After that window, a medical evaluation is required, and if no medical cause is found, it is recorded as a refusal to test.
What cutoff values are used in federal urine drug tests?
For marijuana, the screening cutoff is 50 ng/mL and the confirmatory cutoff is 15 ng/mL. Each drug class has its own HHS-published threshold that labs must apply for federally mandated testing.