Blog
Role of Drug Test Strips: Impact on Screening Accuracy

Every screening decision comes with high stakes for compliance officers and rehabilitation program coordinators. Misunderstandings around drug test strip accuracy and interpretation can lead to costly enforcement errors or wrongful program decisions. Drug test strips are valued for their speed and convenience, but as highlighted by the National Institute on Drug Abuse, their results are only a first step and must be followed by confirmatory lab testing. This guide clarifies common misconceptions, offers operational insights, and outlines key best practices for reliable drug screening.
Drug test strips explained and common misconceptions
Drug test strips are screening tools that use antibody technology to detect drugs or their metabolites in urine samples. Think of them as the first checkpoint in a verification process rather than a final verdict. Compliance officers and rehabilitation coordinators rely on these strips because they deliver results in minutes, making them practical for high-volume screening situations.
Here’s how the chemistry actually works. Antibodies in the strip bind to specific drug molecules or their metabolites when urine contacts the test area. If a match occurs, a color change appears on the strip, signaling a potential positive result. This binding mechanism is precise but not foolproof—similar molecular structures from other substances can sometimes trigger false positives.
Common misconceptions about drug test strips often lead to incorrect interpretations and enforcement errors. Understanding what these misconceptions are protects your organization from liability and ensures fair treatment of individuals being tested.
Key misconceptions to address:
- A positive strip result confirms drug use (it only indicates presence, not consumption timing)
- Test strips never produce false positives (prescribed medications can trigger them)
- Results are definitive without confirmation (they require secondary testing)
- All positive results mean policy violations (metabolites persist for days after use)
- Visual interpretation is always accurate (faint lines create ambiguity)
The research from the National Institute on Drug Abuse confirms that while urine drug tests provide crucial initial screening information, their results must be interpreted carefully and supplemented with more specific testing when necessary. A positive strip indicates only that drug metabolites are present—not whether the person ingested the substance today, yesterday, or three days ago.
Positive urine drug screens are preliminary findings that require confirmatory testing before any personnel decisions are made.
Prescribed medications represent the most common source of misinterpretation. Cold medicines, pain relievers, and antidepressants can cross-react with antibodies designed for other substances. Your testing protocols should account for this reality by collecting medication histories before drawing conclusions.
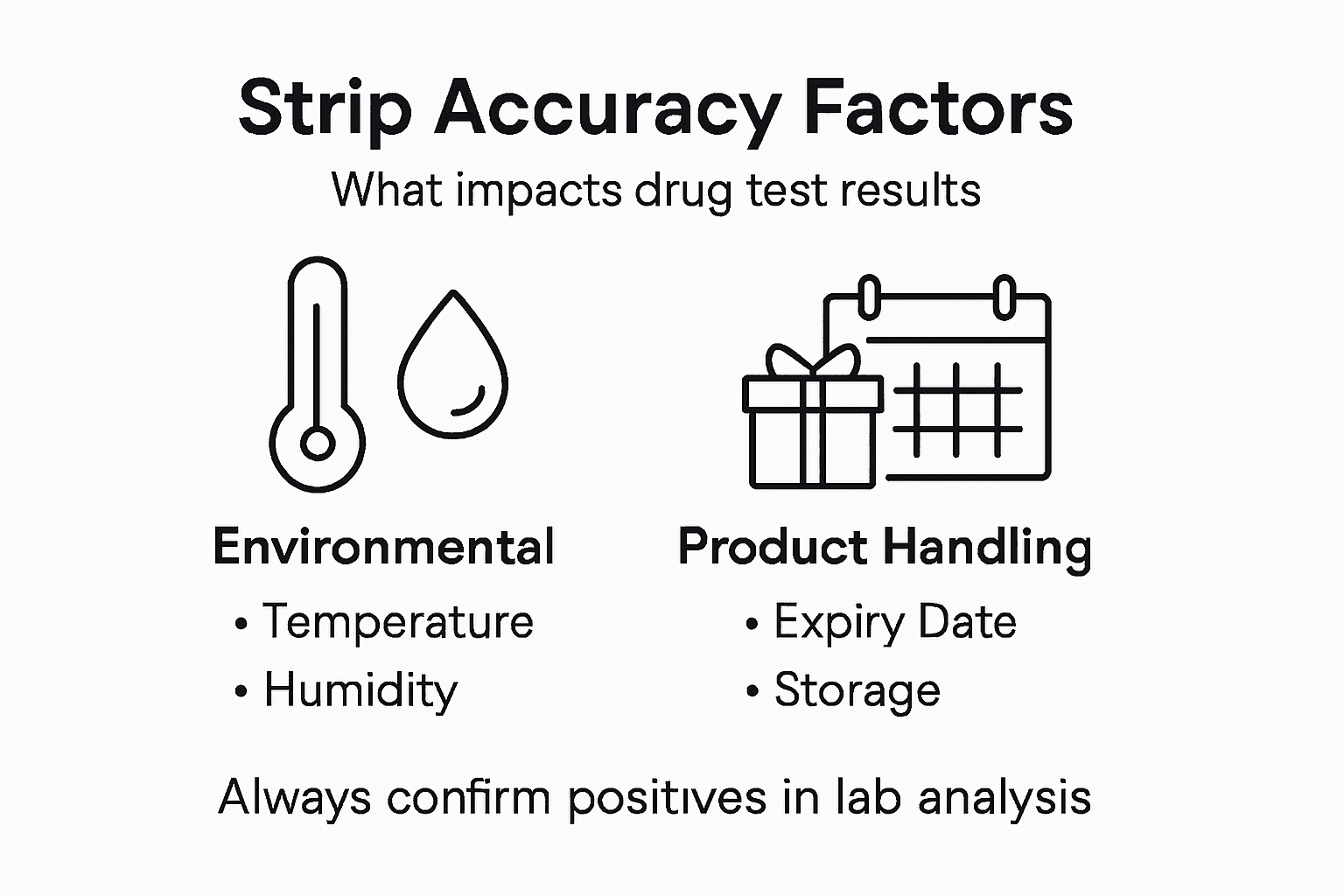
Temperature and storage conditions also affect strip accuracy. Expired strips, improperly stored supplies, or samples outside the acceptable temperature range can produce unreliable results. This is why partnering with providers offering CLIA-waived testing supplies with proper handling documentation matters for your compliance records.
When understanding drug screening terminology and compliance requirements, you’ll notice that industry standards emphasize the distinction between screening and confirmatory testing. Screening strips serve their intended purpose—rapid, cost-effective initial detection—when interpreted correctly within your organizational protocols.
Implement two-step verification processes rather than relying solely on strip results. Confirmatory testing using gas chromatography-mass spectrometry or immunoassay confirmation eliminates ambiguity and protects your agency from wrongful discipline claims.
Pro tip: Establish written protocols requiring all positive strips to undergo confirmatory testing before any action is taken, and ensure staff understand that strip results alone cannot justify employment decisions or program termination.
Types of drug test strips and core differences
Drug test strips come in multiple configurations, each designed to detect specific substances or combinations of drugs. Understanding which strip type serves your operational needs prevents wasted resources and ensures you’re screening for the right compounds. Your choice depends on your agency’s risk profile, budget constraints, and the populations you’re monitoring.
Single-panel strips target one specific drug or drug class at a time. These strips offer maximum simplicity and are ideal when you need focused screening for a particular substance. Use single-panel strips when testing for compliance with specific drug restrictions or monitoring individuals with known substance abuse histories.
Multi-panel strips detect multiple drugs in a single test, typically ranging from 5 to 12 substances per strip. This approach reduces testing time and cost when comprehensive screening matters. Multi-panel strips are standard in workplace compliance programs, rehabilitation intake assessments, and correctional facility monitoring because they capture a broader drug profile with one collection event.
Common drug test strip configurations include:
Here’s a quick comparison of single-panel vs. multi-panel drug test strips:
| Attribute | Single-Panel Strip | Multi-Panel Strip |
|---|---|---|
| Target Substances | One specific drug | 5-12 drugs, broad coverage |
| Cost Efficiency | Lower per test | Better for bulk screening |
| Use Case | Focused compliance | Workplace, rehab, corrections |
| Complexity | Simple interpretation | More training required |
- Single drugs: Marijuana, cocaine, methamphetamine, or PCP detection only
- Five-panel: Cocaine, marijuana, amphetamine, opioids, and PCP
- Ten-panel: Adds benzodiazepines, methadone, barbiturates, and tricyclic antidepressants
- Specialty strips: Fentanyl, synthetic opioids, and emerging drugs like nitazenes
Validation standards matter more than you might think. The National Institute of Standards and Technology collaborates with AOAC International to ensure drug test strips meet consistent sensitivity and specificity benchmarks across manufacturers. This means a validated fentanyl strip from one provider performs comparably to another validated fentanyl strip, giving you reliability across your supply chain.
Emerging synthetic opioids like fentanyl and nitazenes require specialized detection strips validated specifically for these compounds—standard opioid panels may not identify them reliably.
Strip performance varies based on detection thresholds, which are the minimum drug concentrations required to produce a visible positive result. A lower threshold catches more cases but increases false positives from trace metabolites. Higher thresholds reduce false positives but might miss light users or recent exposures. Your threshold choice should align with your program’s enforcement philosophy.
When selecting types of drug test kits suited to your compliance operations, consider whether dip strips, cup tests, or cassette formats fit your workflow. Dip strips require separate cups and visual interpretation, while integrated cup tests combine collection and detection in one device, reducing contamination risks and staff interpretation errors.
Temperature stability and shelf life differ significantly between strip types. Some specialized strips for emerging drugs may have shorter stability windows, requiring more frequent inventory rotation. Budget accordingly and establish expiration tracking systems to prevent using outdated supplies that could compromise accuracy.
Pro tip: Match your strip type to your actual testing volume and substance concerns—buying expensive multi-panel strips for single-drug monitoring wastes money, while buying single-panel strips for comprehensive screening creates staff frustration and hidden costs.
How drug test strips operate in screening
Drug test strips operate through a systematic process that begins with specimen collection and ends with visual result interpretation. Understanding each step helps compliance officers recognize where errors can occur and why confirmatory testing remains non-negotiable. The entire process—from collection to interpretation—takes roughly five to ten minutes, making strips practical for high-volume screening environments.
Specimen collection is the first critical step. Your staff collects urine in a clean container, ensuring proper temperature maintenance and immediate testing or proper storage. Contaminated specimens or samples stored too long produce unreliable results before the strip even enters the picture. This is why chain-of-custody protocols matter at every stage.
Once collected, the specimen gets prepared by bringing it to room temperature if refrigerated. The strip is then inserted into the urine sample, submerging the absorbent pad area for the time specified in manufacturer instructions—typically three to five seconds. The liquid travels upward through the strip via capillary action, carrying drug metabolites toward the detection zones.
The immunoassay detection process works as follows:
- Antibodies on the strip bind to drug metabolites in the specimen
- Colored particles attach to the antibody-drug complex
- The complex moves along the strip toward test and control lines
- Lines develop based on whether metabolites were present
The control line appears on every valid test, confirming the strip functioned properly and the test procedure was executed correctly. If no control line appears, the test is invalid and must be repeated with a fresh strip and new specimen. This single detail prevents invalid results from being misinterpreted as negatives.
The test line indicates whether drug metabolites exceed the cutoff threshold. A visible test line means the result exceeds the cutoff concentration; no line suggests the specimen is negative or below the detection limit. The intensity of the line doesn’t matter—faint or dark lines both indicate positive results above the threshold.
The absence of a test line with a visible control line indicates a negative result, not an invalid test—this distinction matters for your enforcement decisions.
Interpretation requires training to avoid common mistakes. Staff often misread faint lines, misidentify which line is which, or fail to wait the full read time before interpreting results. Standardized interpretation protocols, staff certification programs, and written documentation of every result prevent these errors from affecting your screening accuracy.
When implementing drug screening workflow procedures in your organization, emphasize that strip results remain preliminary. Confirmatory testing using gas chromatography-mass spectrometry or laboratory immunoassay confirms initial positives before any employment decisions or program changes occur. This two-step process protects both your agency and the individuals being tested.
Pro tip: Train staff to document the exact time they read each result and photograph ambiguous results before making calls—this creates accountability and provides evidence if results are later questioned.
Regulatory compliance and legal frameworks
Drug test strip regulations operate at multiple levels—federal, state, and organizational—creating a complex compliance landscape for your agency. Understanding these frameworks prevents legal exposure and ensures your screening practices withstand scrutiny. The regulatory environment is evolving rapidly, particularly around harm reduction tools and workplace testing standards.
Federal regulations establish baseline requirements through agencies like the FDA and Department of Health and Human Services. The FDA generally does not regulate drug-checking products intended for illicit drug detection, but research and validation remain encouraged to ensure effectiveness and quality. This regulatory gap creates flexibility but also responsibility—your agency must validate that supplies meet appropriate standards even when federal oversight is minimal.
Organizations like AOAC International establish performance standards that define sensitivity, specificity, and reliability benchmarks for drug test strips. These standards ensure methodological consistency across manufacturers and jurisdictions. When selecting supplies, verify that products meet AOAC-validated protocols, which provides legal defensibility if results are later challenged in court or administrative hearings.
Key regulatory considerations for compliance officers:
- Workplace testing must comply with state-specific drug testing laws varying significantly by jurisdiction
- Rehabilitation programs follow different standards depending on federal funding sources and licensing requirements
- Correctional facilities operate under separate protocols tied to inmate rights and due process standards
- Data privacy regulations govern how test results are stored, accessed, and shared
State-level restrictions significantly impact your operations. Some states impose strict requirements on who can conduct testing, what must be documented, and how results inform employment or program decisions. Other states allow broader discretion but demand higher accuracy standards. Your legal counsel should review state statutes governing drug screening in your specific context before implementing new protocols.
Legal barriers at state and federal levels can restrict test strip use and distribution, so your organization must understand jurisdiction-specific laws before selecting testing methods or procedures.
Chain-of-custody documentation protects both your agency and individuals being tested. Proper documentation demonstrates that specimens were handled correctly, reducing disputes about result validity. When implementing workplace drug testing compliance procedures, establish written protocols requiring documented verification of specimen integrity, testing procedures, and result interpretation at every step.
Confirmatory testing requirements vary by context. Employment decisions typically require laboratory confirmation of positive strips before termination. Rehabilitation program advancement might allow strips alone for preliminary decisions, but major program changes should still follow confirmatory testing. Know which standard applies in your jurisdiction.
Pro tip: Retain test strips, documentation, and specimen records for the timeframe required by your state law—typically three to seven years—and consult employment counsel about specific retention periods that protect your agency from liability claims.
Practical risks, limitations, and best practices
Drug test strips deliver rapid results but operate within real-world constraints that affect accuracy. Recognizing these limitations protects your organization from costly errors and ensures fair treatment of individuals tested. The key is understanding when strips work and when they fall short.
False positives represent the most common risk. Chemically similar substances can trigger antibodies designed for other drugs, producing positive results without actual drug use. Cold medicines containing pseudoephedrine, certain pain relievers, and antidepressants have documented cross-reactivity patterns. Collecting medication histories before testing helps contextualize unexpected positives.
False negatives occur when drugs present in the specimen fall below detection thresholds or the test panel doesn’t include the substance being used. Someone using fentanyl might test negative on a standard opioid panel because fentanyl requires specialized strips. Synthetic drugs and emerging substances often evade detection from older strip configurations.
Common sources of inaccuracy include:
- Sample adulteration or dilution from attempted cheating
- Environmental factors affecting strip performance or specimen integrity
- Improper storage or expired supplies reducing sensitivity
- Metabolite levels below cutoff thresholds despite recent use
- Visual misinterpretation of faint or ambiguous test lines
Environmental conditions damage strip accuracy significantly. Extreme temperatures, humidity exposure, or improper storage degrade the antibodies and colored particles on strips. Specimens outside acceptable temperature ranges deteriorate chemically. These storage and environmental factors often escape attention but directly impact result reliability.
Below is a summary of key risks and best practices for drug test strip screening:
| Risk Category | Common Risk | Recommended Best Practice |
|---|---|---|
| False Positives | Medication cross-reactivity | Collect medication history |
| False Negatives | Below cutoff/metabolite mismatch | Update strip panels regularly |
| Human Error | Misinterpretation of lines | Train and certify staff |
| Environmental | Storage or temperature issues | Use inventory tracking systems |
False positives and false negatives both occur with strip testing, making confirmatory laboratory testing non-negotiable before any significant personnel or program decisions.
Best practices begin with confirmatory testing protocols. Every positive strip result requires secondary verification using gas chromatography-mass spectrometry or laboratory immunoassay before enforcement action. This two-step approach eliminates ambiguity and protects your agency legally.
Stay current with testing technology advances. Emerging synthetic opioids, designer drugs, and novel substances regularly appear faster than strip technology updates. Subscribe to industry alerts and review your testing panels annually to ensure coverage matches actual substance threats in your population.
Documentation creates accountability and provides evidence if results are later disputed. Record specimen collection time, temperature, testing staff identity, exact read time, result interpretation, and any anomalies observed. Photograph ambiguous results. This documentation trail demonstrates procedural integrity when questioned.
Train staff comprehensively and verify competency regularly. Misinterpretation causes more errors than strip limitations. Certification programs, annual refresher training, and supervised testing sessions maintain staff proficiency and reduce human error by 40 to 60 percent based on compliance data.
Pro tip: Implement a quality assurance program using positive and negative control samples tested weekly to verify strip batches function correctly and staff interpret results accurately before testing actual specimens.
Enhance Your Drug Screening Accuracy With Trusted Supplies
Accurate drug test strips depend on careful selection, proper storage, and reliable detection thresholds outlined in this article. If you have struggled with false positives or uncertain results, your organization is not alone. Common challenges include medication cross-reactivity, environmental factors affecting strip performance, and interpretation errors. These obstacles can create costly compliance risks and unfair outcomes.
At Buy Test Cup, we understand these pain points and offer a wide range of validated drug testing supplies designed to meet your exact screening needs. From single-panel dip strips to comprehensive multi-panel test cups, our products ensure sensitivity and specificity aligned with regulatory standards. Backed by CLIA-waived certifications and supported with detailed handling instructions, you can trust our supplies to deliver rapid, accurate results in any environment.
Ready to reduce false positives and elevate your compliance protocols? Explore our inventory of types of drug test kits suited to your compliance operations and learn how to implement best practices from collection to interpretation with our drug screening workflow guide for accurate results. Don’t let uncertainty slow you down. Visit Buy Test Cup today to secure reliable, cost-effective testing solutions that support your high standards and protect your organization.
Frequently Asked Questions
What are drug test strips and how do they work?
Drug test strips are screening tools that use antibody technology to detect drugs or their metabolites in urine samples. They provide rapid results by binding to specific drug molecules in the specimen and showing a color change based on the presence of drugs.
How accurate are drug test strips in detecting drugs?
While drug test strips offer quick initial screening, they are not foolproof. False positives can occur due to prescribed medications or other substances that chemically resemble the target drugs. Confirmatory testing is crucial to ensure accuracy before making any decisions based on strip results.
What should I do if a drug test strip shows a positive result?
A positive result from a drug test strip indicates the presence of drug metabolites but does not confirm drug use. It is essential to follow up with confirmatory testing, such as gas chromatography-mass spectrometry, before taking any personnel actions or program changes.
What factors can affect the accuracy of drug test strips?
The accuracy of drug test strips can be influenced by several factors, including the quality of the strip, storage conditions, specimen temperature, and human interpretation errors. Adhering to proper storage practices and training staff on correct interpretation can help mitigate these risks.